
Section Abstract Introduction Methods Results Discussion Conflict of Interest Acknowledgment References
Basic Medical Research
X-ray radiation effect of C-arm on adipose tissue-mesenchymal stem cell viability and population doubling time
pISSN: 0853-1773 • eISSN: 2252-8083
http://dx.doi.org/10.13181/mji.v25i1.1335 Med J Indones. 2016;25:10–8
Received: December 23, 2015
Accepted: March 16, 2016
Author affiliation:
1 Department of Orthopaedic and Traumatology, Faculty of Medicine, Universitas Indonesia, Cipto Mangunkusumo Hospital, Jakarta, Indonesia
2 Stem Cell Medical Technology Integrated Service Unit, Cipto Mangunkusumo Central Hospital, Faculty of Medicine Universitas Indonesia, Jakarta, Indonesia
3 Stem Cell and Tissue Engineering Cluster, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia
4 Department of Histology, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia
Corresponding author:
Ahmad J. Rahyussalim
E-mail: rahyussalim71@ui.ac.id
Background
Adipose tissue derived mesenchymal stem cells (AT-MSCs) are relatively easy in isolation procedure compared to bone marrow-derived. Minimally invasive MSC injections need C-arm as guidance that potentially influence the cell viability and doubling time. This study aimsed to determine the effect of C-arm X-ray exposure on AT-MSC viability and population doubling time (PDT).
Methods
This experimental study used cryopreserved adipose tissue derived MSCs stored in Stem Cell Medical Technology Integrated Service Unit Cipto Mangunkusumo Hospital. Cells were thawed, propagated, and exposed to varying doses of C-arm X-ray radiation. Stem cell viability was measured, and then the cells were cultured to assess their PDT. Generalized linear models test was used to compare cell viability between post-thaw, post-propagation, post-radiation, post-culture post-radiation, and control and between radiation dose groups. Kruskal-Wallis test assessed PDT between various radiation doses in post-radiation groups. Wilcoxon test was used to assess PDT between preradiation and post-radiation groups.
Results
Mean confluence period of adipose MSCs postirradiation was 4.33 days. There was no statistically significant difference in MSC viability after X-ray exposure between pre- and post-irradiation groups (p=0.831). There was no correlation between post-irradiation viability and radiation dose (p=0.138, r=0.503). There were no significant differences in PDT between pre- and post-culture postirradiation groups and between various radiation doses in post-irradiation groups (p=0.792).
Conclusion
MSC viability and PDT were not influenced by radiation exposure up to 32.34 mgray.
Keywords
AT-MSCs, population doubling time, irradiation, viability
Stem cells are regarded as future treatment for a variety of diseases due to their differentiation, proliferation, and regeneration capability. Mesenchymal stem cell (MSC) is a type of stem cell, which can be found in bone marrow, adipose tissue, umbilical cord, brain, liver, muscle, skin, and embryo.1–5 Although MSCs can be found in many kinds of tissues and organs, they constitute only one among 106 adult bone marrow stromal cells, and only one among 104 umbilical cord cells.1 A study by Lubis et al2 reported that MSCs, which were collected from iliac crest could be cultured in vitro so that they could meet the need for cell therapy. Adipose tissue-mesenchymal stem cells (AT-MSCs) are increasingly utilized in clinical practice. Compared to other types of MSCs, AT-MSCs are relatively easy in isolation procedure, can be obtained less painful from various sites (e.g. abdomen, thigh, upper arm, back, yellow marrow, etc), and have similar regenerative and differentiation ability as bone marrow MSCs.5–7 Moreover, for allogeneic use, ATMSCs may be obtained from waste of liposuction. However, AT-MSCs sensitivity to irradiation has not been reported yet.
In orthopedic clinical practice, stem cells are utilized for the treatment of segmental bone defect, articular cartilage and tendon local defect, spinal fusion, and neuronal regeneration.1,2,7 Direct implantation of MSCs for spinal cord injury was relatively safe and may improve neurological deficit.8–11 Intraspinal implantation of MSCs can be done either by open surgery 12 or by minimally invasive technique using C-arm image intensifier that causes X-ray radiation in Cipto Mangunkusumo Hospital. Minimally invasive intraspinal implantation procedure mimics vertebroplasty procedure in term of C-arm guided to reach the lesion or spinal cord as an implantation target.13 Intraspinal implantation in conjunction with open surgery is applied especially for acute injury patient needing immediate surgery. For this case, allogeneic MSCs implantation is a treatment of choice because the cells are available in a laboratory. Minimal invasive intraspinal implantation is applied for chronic or delayed cases without canal problem. It this procedure is done in operating theatre with C-arm imaging intensifier. X-ray image guides the needle to put cells into the lesion precisely. This process it is very important for the cells to directly spreading spinal cord damage without repair the systemically throughout the human body.
A study showed that ionizing radiation up to 10 Gy did not alter functional characteristics and marker expression of MSCs.14 However, Kurpinski et al15 suggested that 0.1 Gy of X-ray may inhibit MSC cycle without interfering osteogenic differentiation in vitro. Irradiation was also found to inhibit stem cell proliferation and differentiation at two weeks post-exposure.16
Laboratories that produced MSCs are now available in Indonesia and the researchers have capabilities to do MSC isolation and culture as well. However, there was no study, which addressed the viability and proliferation capability of AT-MSCs after X-ray radiation exposure of C-arm image intensifier. The present study aimed to evaluate viability and proliferation ability of MSCs after X-ray radiation exposure of the machine.
METHODS
The study design was in vitro laboratory experimental study held in Cipto Mangunkusumo Hospital in February 2015. This study was approved by Ethical Committee for Medical Research of Faculty of Medicine Universitas Indonesia-Cipto Mangunkusumo Hospital with No. 164/UN2.F1/ ETIK/II/2015.
This research used cryopreserved AT-MSCs. The cells were derived from a healthy donor, a 19-yearold girl who had an open reduction and internal fixation in September 2014. With patient’s consent, some adipose tissue was harvested from femoral region by surgery procedure, processed, and the cells were cultured in two cycles before finally were cryopreserved. The inclusion criteria were the cells should expressed positive CD73 and CD90 markers, and were negative for CD34 marker. The cells were indeed mesenchymal stem cells as was the criteria of International Society for Cell Therapy (ISCT). Exclusion criteria were if there was not enough cells obtained for one batch of experiment and cultures were contaminated during experiment. Sample size (n) was set three for each group.
AT-MSC samples were taken from tissue bank at Stem Cell Medical Technology Integrated Service Unit, Cipto Mangunkusumo Hospital, CMU 2 Building, 5th floor, Jl. Diponegoro 71, Jakarta, Indonesia.
Preparation of AT-MSCs
Cryopreserved AT-MSCs were thawed, and washed in complete medium (1% penicillin/ streptomycin, 1% amphotericin B, 1% heparin (1,000 U), 1% glutamax-L, and 10% platelet rich plasma containing α-MEM) and were propagated in T25 flasks, and when confluence was reached, MSCs were harvested using TrypLE Select, neutralized by an equal amount of complete medium, and viability was directly assessed using trypan blue exclusion method. Viability was calculated as percentage ratio of the number of living and total cells. Furthermore, population doubling time (PDT)17,18 was calculated.
C-arm X-Ray exposure
Mesenchymal stem cells were divided into ten groups (one control group and nine treatment groups), which were put in Eppendorf tubes that contained 1x105 cells/100 μL medium. MSCs of treatment groups were exposed to X-ray from the C-arm image intensifier (Siemens Siremobil Compact L X-ray tube). First group was the control group that did not receive any X-ray exposure. Group II, III, and IV received 50 kV of X-ray exposure for 60, 120, and 240 seconds respectively. Group V, VI, and VII received 60 kV of X-ray exposure for 60, 120, and 240 seconds respectively. Group VIII, XI, and X received 70 kV of X-ray exposure for 60, 120, and 240 seconds respectively. X-ray exposure in each group was repeated three times. X-ray exposure on MSCs was measured by TLD100-H dosimeter, and the dose was interpreted by Harshaw TLD 3500. After X-ray exposure, all groups were evaluated for their viabilities. All experiments were done in duplo.
Mesenchymal stem cells were then cultured in complete medium at 37°C with 5% CO2. The cells were observed and the medium was renewed every two days. When the cells in control group were in 80 to 90% confluent, all cells in all groups were harvested, viability was assessed and PDT was calculated.
Population doubling time measurement
Population doubling time (PDT) was calculated using the equation:
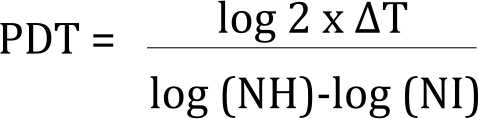
NH= harvested cell number
NI= cell number at seeding
Δt = time from seeding to harvesting (in days)
Data collection and analysis
Data were collected at pre-irradiation, postirradiation, and post-irradiation-and-culture to evaluate viability and PDT for all groups. Statistical analyses were done with SPSS version 17.0 by generalized linear model to evaluate differences in viability between study groups. Spearman test was used to calculate correlation between pre-irradiation, postirradiation, and post-irradiation-and-culture viability for all groups. One-way Anova test or Kruskal-Wallis test as alternative was used to evaluate the significanct difference of post-irradiation population doubling time between study groups. Pearson test (if data distribution was normal) or Wilcoxon test (if data distribution was abnormal) was used to evaluate the significanct difference between pre-irradiation and post-irradiation population doubling time between groups.
RESULTS
AT-MSCs post-culture confluent at day four postculture can be seen in Figure 1. Viability showed slight difference between X-ray exposure group (Figure 1 B, 1C and 1D) compared to control group (Figure 1A). Before the cells were harvested, we had observed a retraction in cultured AT-MSCs with exposure strength of 50 kVp, at all exposure duration (Group II, III, and IV), which can be seen in Figure 2D. Table 1 shows the mean values of viability and PDT at various irradiation doses and control at pre-irradiation, post-irradiation, and postirradiation- and-culture. Mean difference due to irradiation waiting time in control group was 5.07% while mean difference of AT-MSCs postexposure viability for all groups was 7.61%.
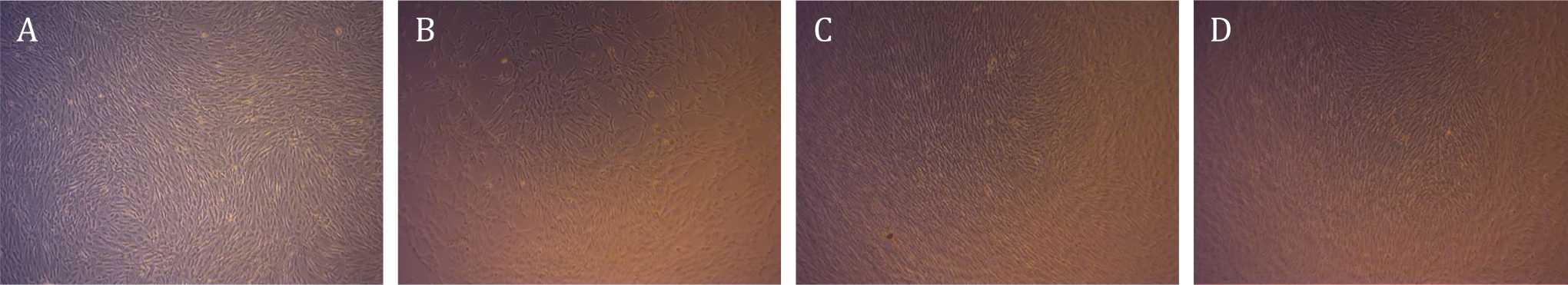
Figure 1. Adipose tissue-mesenchymal stem cells post-culture confluent at day four post-culture. Viability showed slight difference between X-ray exposure group (B, C, and D) compared to control group (A). There was no statistically significant diffference pertaining to viability among the groups (magnification 400x)
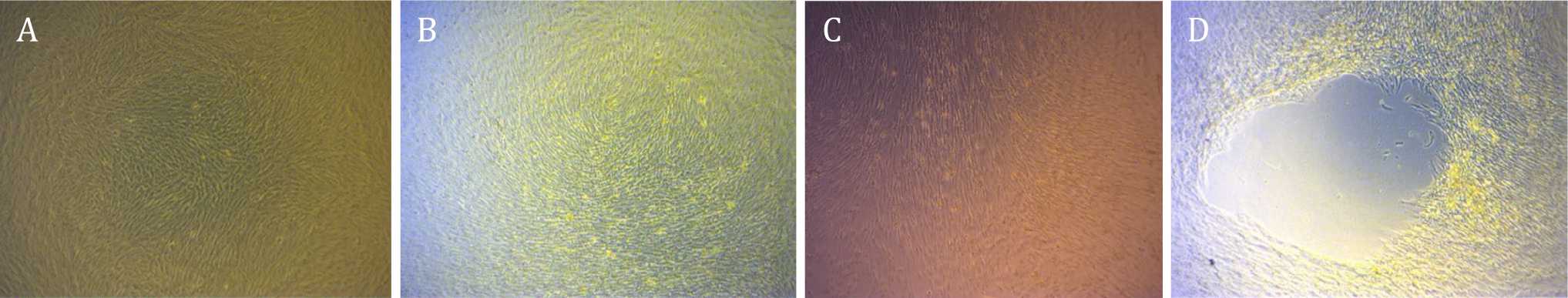
Figure 2. Adipose tissue-mesenchymal stem cells post-culture showed confluent cells post-thawing (A, B, and C). Retraction formation due to over confluence was shown by cells post-culture after radiation (D) (magnification 400x)
Table 1. Mean values of viability and PDT of the study groups at pre-irradiation, post-irradiation, and post-irradiation-and-culture
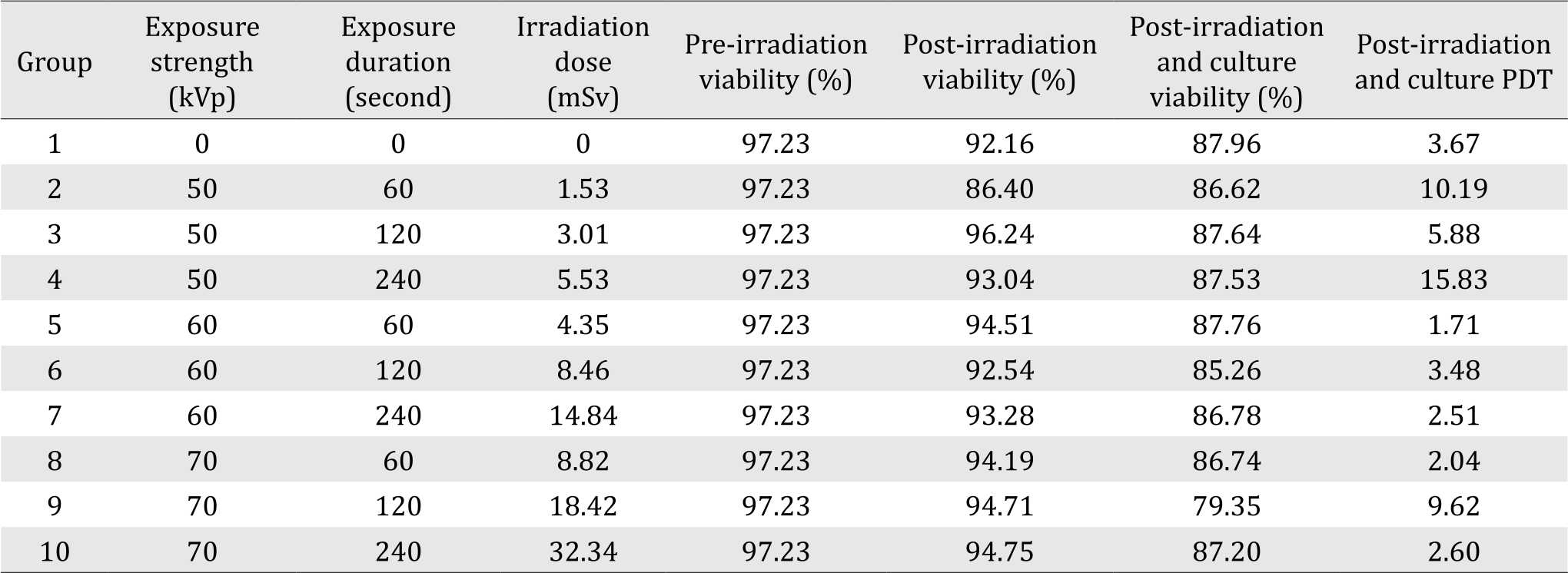
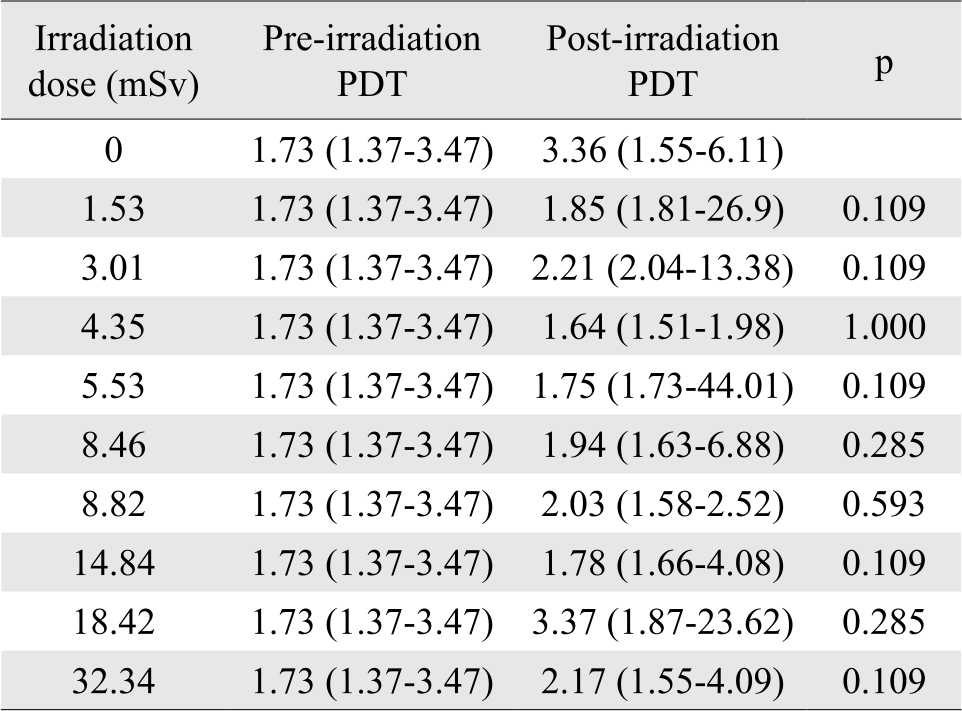
The shortest post-irradiation-and-culture PDT was 2.04 days, found at 8.82 mSv dosage (group VIII); the longest PDT was 15.83 days, found at 5.53 mSv dosage (group IV). Mean postirradiation- and-culture PDT for experimental groups was 38% longer than that of control group. There was no significant difference between viability values at pre-irradiation, post-irradiation, and post-irradiation-andculture (p=0.831) (Figure 1). There was correlation between post-irradiation viability and irradiation dose, but it was not statistically significant (r=0.503; p=0.138). Table 2 shows median, minimum, and maximum values of PDT at various irradiation doses, at pre-and post-irradiation. There was no statistically significant difference between pre-irradiation and post-irradiation PDT in all experimental groups. Moreover, there was no statistically significant difference of post-irradiation population doubling time between irradiation doses (p=0.792).
Table 2. Median, minimum and maximum values of PDT at various irradiation doses, at pre-and post-irradiation
DISCUSSION
In this study, we found a correlation between postirradiation- and-culture viability and irradiation dose (r=-0.648, p=0.043). We also found a correlation between post-irradiation viability and irradiation dose, although it was not statistically significant. From those findings, we suspected that some DNA was damaged due to irradiation, but cell death did not occur until the attempt to repair the damage was failed. Some DNA repair mechanism occurs at mitosis; therefore, we found a decrease in post-irradiation-and-culture viability. Viability represents stem cell ability to withstand external stressors. AT-MSCs are sensitive to irradiation. Islam et al19 suggested that irradiation impaired bone marrow stem cell mitotic and differentiation capability, cell cycle, and gene expression. The reduced viability may increase the amount of MSCs required in clinical practice. The present study showed a tendency of decrease in cell viability after X-ray exposure, although it was not statistically significant. There are several contributing factors to cell viability. Sensitivity to irradiation is affected by cell ability to overcome irradiation related genome damage. In other words, failure to repair post-irradiation cell damage may lead to reduce proliferation ability and cell death.14
The effect of irradiation to MSCs depends on various factors, including radiation dose, cell/tissue endogenous, and environmental factors. Irradiation affects stem cell through deoxyribonucleic acid (DNA) damage, cell cycle impairment, senescence, and cell death related to genetic and epigenetic alteration.20 Cell and tissue injury due to irradiation is caused either by direct macromolecule ionization or by some indirect processes through free radicals from water radiolysis.19 Ionization and reactions with free radicals may alter structure and function of DNA, lipid, and protein. Those changes may impair cell metabolic function that lead to cell injury or even cell death. DNA is the most important target macromolecule in cell injury and death.21 Free radicals may enter the cell by diffusion through cell membrane and thereby damaging cell macromolecules, particularly DNA. Cell endurance to irradiation is affected by the ability to produce antioxidants and the ability to repair DNA damage.22 There are two mechanisms of DNA repair: non-homologous end joining (NHEJ) and homologous recombination (HR). The NHEJ has six components: Ku70, Ku80, DNA-PK, XRCC4, DNA ligase IV (LigIV), and XRCC4-like factor (XLF). In NHEJ process, DNA damage is identified by Ku proteins; which bind and activate DNA protein kinase. Then end-processing enzymes, polymerase, and DNA ligase IV are called upon and activated. Moreover, HR begins with ssDNA generation. The HR process is affected by MRE11- Rad50-NBS1 (MRN) protein complex. Catalyzed by Rad51, BRCA1, and BRCA2, ssDNA is processed by polymerase, nuclease, helicase and DNA ligase. HR may reintroduce replication process and repair inter-strand DNA crosslink. While NHEJ may occur at all cell cycle, HR can only occur at S and G2 phase.23
Li et al24 reported that high radiation dose substantially reduced the ability to differentiate, although it did not diminish completely. Furthermore, Pawlik et al25 reported that cell sensitivity to irradiation varied according to its cell cycle; the cell was relatively sensitive during M and G2 phase, and was relatively resistant at the end of S phase. Our study did not check the cell cycle phase, and this fact was the limitation of our study.
Radiation dose unit used in this study is Sievert. Sievert is an international unit of exposure dose or equivalent dose, which is equal to received dose (gray) multiplied by quality factor. Quality factor for X-ray is 1, so 1 sievert = 1 gray. Irradiation dose at the present study was between 1.53-32.34 mSv. Upon the radiation exposure, viability of MSCs was decreased. In accordance with Nicolay et al14, human MSCs were less radiosensitive than cells that had already differentiated into primary fibroblasts. Irradiation as low as 0.1 gray may inhibit MSC cycle without affecting its osteogenic differentiation in vitro.16 Other study reported that low-dose irradiation may improve osteoblastic differentiation and mineralization in vitro.24 We did not explore the differentiation ability of MSCs after irradiation exposure. This study was not along with data from Liang et al26 that which showed low-dose 50 and 75 mGy radiation improved MSC proliferation compared to control and 100 mGy radiation, due to transition between G1 and S phase of cell cycle.26 Higher dose of radiation at 2-4 cGy caused DNA damage with subsequent transduction signal pathway activation to repair the damage. Moreover, high-dose 4 Gy irradiation caused DNA damage that was followed by cell apoptosis and proliferation.19,27 However, according to Singh et al22 radiation exposure in vivo up to 9 Gy did not affect proliferation ability. Furthermore, Kurpinski et al15 suggested that MSCs responded well to radiation exposure by repairing DNA damage.
Mesenchymal stem cells viability in the present study decreased in the majority of experimental group, including all post-irradiation-andculture groups. The decreased viability of postirradiation- and-culture groups may be related to senescence of the MSCs itself. Schallmoser et al28 suggested that aging MSCs had impaired cell proliferation, altered phenotype, gene expression, and cell nucleus structure, as well as impaired protein processing and cell metabolism. However, this suggestion was not supported by Zuk et al29 which showed that MSCs were relatively stable, with a senescent rate of 5% at the 10th passage and only 15% at the 15th passage, but the MSCs used in Zuk et al29 study were not cryopreserved, while we used cryopreserved cells which were shown to undergo senecence at lower passages compared to fresh cells.30
Pre-irradiation PDT in the present study was in accordance with Mitchell et al31 which reported that MSC PDT at the first to fourth passage was 86.6-112.8 hours. In contrast with However, different with our study, Zhu et al32 reported that MSC PDT at the third passage was 36 hours, and Wall et al33 reported that until the fifth passage PDT was 45 hours. At the present study, mean post-irradiation PDT was higher than mean pre-irradiation PDT, but did not differ significantly. Pre-irradiation and postirradiation AT-MSCs were in different passage due to culturing. A study by Gruber et al34 suggested that PDT increased significantly in line with the period of passage. In our study, a small passage difference (only one period of passage) and small radiation exposure were not enough to produce a statistically significant lengthening of PDT. The findings were also in accordance with Peng et al.35
With doses of 5.53 mSv (group IV) and 18.42 mSv (group IX), PDT was dramatically increased. Those phenomena were not found in any other groups, although all MSCs underwent the same process of thawing. The substantial increase of PDT in groups IV and IX may be caused by difference in cell cycle when exposed to irradiation, or difference in the location of DNA damage. This is one of the weaknesses of our study as we did not specify at which cell cycle the irradiation was done nor the location of DNA damage, as DNA damage due to X-ray radiation usually occurs at random.
Hahn et al36 found a correlation between prolonged PDT and increase in relative resistance to radiation, the lengthening of PDT permitted the cells to repair DNA damage. Nicolay et al14 admitted that cell failure to repair the radiationrelated damage may lead to loss of proliferation ability or cell death. This may decrease the initial number of proliferation capable cells, thereby may prolong the confluence time and PDT. MSCs may retain post-irradiation proliferation ability with x-ray exposure up to 10 Gy. However, MSC PDT may be prolonged up to three times compared to primary fibroblasts.
Mesenchymal stem cells proliferation rate was influenced by several factors such as cell source, culture medium composition and quality, such as supplement (FBS, platelet lysate), glucose and glutamine concentration, presence of hypoxia, cell density, flask size, plastic quality, and addition of growth factors. We used α-MEM as basal medium to support ATMSC proliferation. Similar to Suryani et al study18 we also used 10% platelet lysate since this was better and more economical compared to a commercial medium (Mesencult®) at the first to fifth passage, although in the initial culture Mesencult® was better.
Through an in vitro study, Sardjono et al37 found spontaneous differentiation and senescence of ATMSCs after six passages. They suggested that cell viability may decrease after washing process by PBS. They washed the cells in PBS before cell count to minimize trypsin effect that was used to separate MSCs from the plastic container. Therefore we did not wash the cells as we used tryple Select, which was milder compared to trypsin, in accordance with Pawitan et al38 to minimize the confounding factors.
The duration of radiation exposure was based on C-arm image intensifier usage in clinical practice at our institute, especially for patients with spinal orthopaedic surgery. C-arm at our institution was used for one to three minutes; it was in accordance to Boszczyk et al13 in which kyphoplasty assumed X-ray exposure from C-arm for one to two minutes.
The TLD was used to estimate the dose received by AT-MSCs from C-arm image intensifier. Compared to other types of dosimeter, TLD was more sensitive, more durable, more precise (measure X-ray exposure below 1 mGy), more responsive, and more consistent (unaffected by humidity nor magnetic field).39 Moreover, TLD 100-H that was used in this study was a dosimeter of choice for measuring low-dose X-ray exposure in clinical practice.40-42
In this study, we observed a retraction in cultured AT-MSCs with exposure strength of 50 kVp, at all exposure duration (group two, three, and four) (Figure 2). Retraction at the study of Pawitan et al38 was found as a result of confluence imbalance in the flask; monolayer at the flask periphery and over confluence parts grew upwards, forming multiple layers, due to prolonged culture. These multilayer cells were clearly seen at the beginning, but subsequently became thickened and formed micromasses with blurred appearance due to formation of chondrogenic matrix. We suggested that the retraction in our study represented differentiated cells with a decrease in proliferation ability, thereby lengthen MSC PDT of corresponding study groups. Moreover, differentiated cells were not able to proliferate anymore, leading to a decrease in their viability. This fact was one of the limitations in our study, which might have a consequence on the results.
Age was a contributing factor to biological properties of MSCs, but we supposed that it did not affect our study significantly. MSCs in this study were harvested from a 19-year-old girl. According to Gruber et al34 MSCs collected from old donors (mean age of 56 years old) showed a longer PDT at the first to fourth passage. In addition, Zhu et al32 reported a decrease in MSC proliferation ability, which correlated well with increasing age. In contrary, Schipper et al43 did not find any significant difference of cell proliferation between three age groups.
Overall, the limitations of our study included the fact that the cells were harvested from a single young donor, and might show different results on older donor. Moreover, we did not check the cell cycle and senescensce profile, and some of the cultures were grown to overconfluence. Therefore, further studies are needed to verify our results.
For future directions, we suggest to use cells below passage 10 for fresh cells and for cryopreserved cells below passage five, which are not over confluence to reduce the presence of differentiated and senescent cells. Moreover, as cells from logaritmic phase (in mitotic phase) are more prone to irradiation damage, and as viability and PDT were not influenced by radiation exposure up to 32.34 mGy, we recommend to use radiation exposure up to 32.34 mGy.
In conclusion, MSC viability and PDT were not influenced by radiation exposure up to 32.34 mGy.
Conflicts of Interest
The authors affirm no conflict of interest in this study.
Acknowledgment
This study was funded by the research grant from Directorate of Research and Public Service University of Indonesia (Hibah Pasca- Sarjana UI 2015), contract No. 1689/UN2.R12/ HKP.05.00/2015.
REFERENCES
- Barry FP, Murphy JM. Mesenchymal stem cells: clinical applications and biological characterization. Int J Biochem Cell Biol. 2004;36(4):568–84.
- Lubis AM, Sandhow L, Lubis VK, Noor A, Gumay F, Merlina M, et al. Isolation and cultivation of mesenchymal stem cells from iliac crest bone marrow for further cartilage defect management. Acta Med Indonesia. 2011;43(3):178–84.
- Wang Y, Han Z, Song Y, Han ZC. Safety of mesenchymal stem cells for clinical application. Stem Cells Int. 2012;2012:652034.
- Hu L, Hu J, Zhao J, Liu J, Ouyang W, Yang C, et al. Side-byside comparison of the biological characteristics of human umbilical cord and adipose tissue-derived mesenchymal stem cells. Biomed Res Int. 2013;2013:1–12.
- Sachs PC, Francis MP, Zhao M, Brumelle J, Rao RR, Elmore LW, et al. Defining essential stem cell characteristics in adipose-derived stromal cells extracted from distinct anatomical sites. Cell Tissue Res. 2012;349(2):505–15.
- Pawitan JA. Prospect of adipose tissue derived mesenchymal stem cells in regenerative medicine. Cell Tissue Transplant Ther. 2009;2:7–9.
- Haddad NE. Mesenchymal stem cells: immunology and therapeutic benefits. In: Gholamrezanezhad DA, ed. Stem Cells in Clinic and Research. InTech. 2011. http:// www.intechopen.com/books/stem-cells-in-clinic-andresearch/ mesenchymal-stem-cells-immunology-andtherapeutic- benefits.
- Park HC, Shim YS, Ha Y, Yoon SH, Park RS, Choi BH, et al. Treatment of complete spinal cord injury patients by autologous bone marrow cell transplantation and administration of granulocyte-macrophage colony stimulating factor. Tissue Eng. 2005;11(5-6):913–22.
- William JB, Prabakaran R, Ayyappan S, Puskhinraj H, Rao D, Manjunath SR, et al. Functional recovery of spinal cord injury following application of intralesional bone marrow mononuclear cells embedded in polymer scaffold? two year follow-up in a canine. J Stem Cell Res Ther. 2011;1(110):1–5.
- Neirinckx V, Cantinieaux D, Coste C, Rogister B, Franzen R, Wislet-Gendebien S. Concise review: spinal cord injuries: how could adult mesenchymal and neural crest stem cells take up the challenge. Stem Cells. 2014;32(4):829–43.
- Pawitan JA, Damayanti L, Bustami A, Swantari NM. Detection of morphological changes in adipose tissuederived stem cells after passage by the simple spot method. J US-China Med Sci. 2011;8(2):92–8.
- Yoon SH, Shim YS, Park YH, Chung JK, Nam JH, Kim MO, et al. Complete spinal cord injury treatment using autologous bone marrow cell transplantation and bone marrow stimulation with granulocyte macrophagecolony stimulating factor: Phase I/II clinical trial. Stem Cells. 2007;25(8):2066–73
- Li YY, Huang TJ, Cheng CC, Wu MH, Lee CY. Comparing radiation exposure during percutaneous vertebroplasty using one- vs two-fluoroscopic technique. BMC Musculoskelet Disord. 2013;14:38.
- Nicolay NH, Sommer E, Lopez R, Wirkner U, Trinh T, Sisombath S, et al. Mesenchymal stem cells retain their defining stem cell characteristics after exposure to ionizing radiation. Int J Radiat Oncol Biol Phys. 2013;87(5):1171–8.
- Kurpinski K, Jang DJ, Bhattacharya S, Rydberg B, Chu J, So J, et al. Differential effects of x-rays and high-energy 56Fe ions on human mesenchymal stem cells. Int J Radiat Oncol Biol Phys. 2009;73(3):869–77.
- Xu W, Xu L, Chen M, Mao YT, Xie ZG, Wu SL, et al. The effects of low dose x-irradiation on osteoblastic MC3T3-E1 cells in vitro. BMC Musculoskeletal Disord. 2012;13:94.
- Cristofalo VJ, Allen RG, Pignolo RJ, Martin BG, Beck JC. Relationship between donor age and the replicative lifespan of human cells in culture: a reevaluation. Proc Natl Acad Sci U S A. 1998;95(18):10614–9.
- Suryani D, Pawitan JA, Lilianty J, Purwoko RY, Liem IK, Damayanti L. Comparison of fetal bovine serum and platelet-rich plasma on human lipoaspirate-derived mesenchymal stem cell proliferation. Med J Indones. 2013;22(3):146–51.
- Islam MS, Stemig ME, Takahashi Y, Hui SK. Radiation response of mesenchymal stem cells derived from bone marrow and human pluripotent stem cells. J Radiat Res. 2015;56(2):269–77.
- Manda K, Kavanagh JN, Buttler D, Prise KM, Hildebrandt G. Low dose effects of ionizing radiation on normal tissue stem cells. Mutat Res Rev Mutat Res. 2014;761:6–14.
- Shao L, Luo Y, Zhou D. Hematopoietic stem cell injury induced by ionizing radiation. Antioxid Redox Signal. 2014;20(9):1447–62.
- Singh S, Kloss FR, Brunauer R, Schimke M, Jamnig A, Greiderer-Kleinlercher B, et al. Mesenchymal stem cells show radioresistance in vivo. J Cell Mol Med. 2012;16(4):877–87.
- Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461(7267):1071–8.
- Li J, Kwong DL, Chan GC. The effects of various irradiation doses on the growth and differentiation of marrow-derived human mesenchymal stromal cells. Pediatr Transplant. 2007;11(4):379–87.
- Pawlik TM, Keyomarsi K. Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys. 2004;59(4):928–42.
- Liang X, So YH, Cui J, Ma K, Xu X, Zhao Y, et al. The lowdose ionizing radiation stimulates cell proliferation via activation of the MAPK/ERK pathway in rat cultured mesenchymal stem cells. J Radiat Res. 2011;52(3):380–6.
- Ding LH, Shingyoji M, Chen F, Hwang JJ, Burma S, Lee C, et al. Gene expression profiles of normal human fibroblasts after exposure to ionizing radiation: a comparative study of low and high doses. Radiat Res. 2005;164(1):17–26.
- Schallmoser K, Bartmann C, Rohde E, Bork S, Guelly C, Obenauf AC, et al. Replicative senescence-associated gene expression changes in mesenchymal stromal cells are similar under different culture conditions. Haematologica. 2010;95(6):867–74.
- Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211–28.
- Goei N, Liem IK, Pawitan JA, Mediana D. Effect of platelet rich plasma on post cryopreservation viability, morphology, and proliferation of human umbilical cord stem cells. Online J Biol Sci. 2015;15:42–8.
- Mitchell JB, McIntosh K, Zvonic S, Garrett S, Floyd ZE, Kloster A, et al. Immunophenotype of human adiposederived cells: temporal changes in stromal-associated and stem cell-associated markers. Stem Cells. 2006;24(2):376–85.
- Zhu Y, Liu T, Song K, Fan X, Ma X, Cui Z. Adipose-derived stem cell: A better stem cell than BMSC. Cell Biochem Funct. 2008;26(6):664–75.
- Wall ME, Bernacki SH, Loboa EG. Effects of serial passaging on the adipogenic and osteogenic differentiation potential of adipose-derived human mesenchymal stem cells. Tissue Eng. 2007;13(6):1291–8.
- Gruber HE, Somayaji S, Riley F, Hoelsher GL, Norton HJ, Ingram J, et al. Human adipose-derived mesenchymal stem cells : serial passaging, doubling time and cell senescence. Biotech Histochem. 2012;87(4):303–11.
- Peng L, Jia Z, Yin X, Zhang X, Liu Y, Chen P, et al. Comparative analysis of mesenchymal stem cells from bone marrow, cartilage, and adipose tissue. Stem Cells Dev. 2008;17(4):761–73.
- Hahn GM, Bagshaw MA. Serum concentration: effects on cycle and x-ray sensitivity of mammalian cells. Science. 1966;151(3709):459–61.
- Sardjono CT, Setiawan M, Frisca F, Saputra V, Aniko G, Sandra F, et al. Application of a modified method for stem cell isolation from lipoaspirates in a basic lab. Med J Indones. 2009;18(2):91–6.
- Pawitan JA, Suryani D, Wulandari D, Damayanti L, Liem IK, Purwoko RY. Prolonged culture in FBS and FBSsubstitute containing media: spontaneous chondrogenic differentiation of adipose tissue derived mesenchymal stem cells. Int J Pharm Tech Res. 2014;6(1):224–35.
- Olko P, Currivan L, van Dijk JW, Lopez MA, Wernli C. Thermoluminescent detectors applied in individual monitoring of radiation workers in Europe -- a review based on the EURADOS questionnaire. Radiat Prot Dosimetry. 2006;120(1-4):298–302.
- Sofyan H. Keunggulan dan kelemahan dosimeter luminesensi sebagai dosimetri personal dalam pemantauan dosis radiasi eksternal. In: Jakarta: PTKMR BATAN, UI, Kemenkes RI. 2012;1–16. Indonesian.
- Freire L, Calado A, Cardoso JV, Santos LM, Alves JG. Comparison of LiF (TLD-100 and TLD-100H) detectors for extremity monitoring. Radiat Meas. 2008;43(2–6):646–50.
- Sofyan H. Perbandingan tanggapan dosimeter termoluminisensi lif:mg,ti dan lif:mg,cu,p terhadap dosis dalam aplikasi medik. Indones J Nucl Sci Technol. 2012;13:109–18. Indonesian.
- Schipper BM, Marra KG, Zhang W, Donnenberg AD, Rubin JP. Regional anatomic and age effects on cell function of human adipose-derived stem cells. Ann Plast Surg. 2008;60(5):538–44.
Copyright @ 2016 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id