
Section Abstract Introduction Methods Results Discussion Conflict of Interest Acknowledgment References
Clinical Research
Risk factors of sepsis after open congenital cardiac surgery in infants: a pilot study
pISSN: 0853-1773 • eISSN: 2252-8083
http://dx.doi.org/10.13181/mji.v25i3.1450 Med J Indones. 2016;25:182–9
Received: May 17, 2016
Accepted: August 19, 2016
Author affiliation:
1 Department of Surgery, National Cardiovascular Centre Harapan Kita, Jakarta, Indonesia
2 Department of Thoracic and Cardiovascular Surgery, Faculty of Medicine, Universitas Indonesia, Cipto Mangunkusumo Hospital, Jakarta, Indonesia
3 Research and Development Division, National Cardiovascular Centre Harapan Kita, Jakarta, Indonesia
4 Department of Internal Medicine, Faculty of Medicine, Universitas Indonesia, Cipto Mangunkusumo Hospital, Jakarta, Indonesia
Corresponding author:
Dicky Fakhri
E-mail: difawi@yahoo.com
Background
Postsurgical sepsis is one of the main causes of the high mortality and morbidity after open congenital heart surgery in infants. This study aimed to evaluate the role of cardiopulmonary bypass duration, thymectomy, surgical complexity, and nutritional status on postsurgical sepsis after open congenital cardiac surgery in infants.
Methods
A total of 40 patients <1 year of age with congenital heart disease, Aristotle Basic Score (ABS) ≥6 were followed for clinical and laboratory data before and after surgery until the occurrence of signs or symptoms of sepsis or until a maximum of 7 days after surgery. Bivariate analyses were performed. Variables with p≤0.200 were then included for logistic regression.
Results
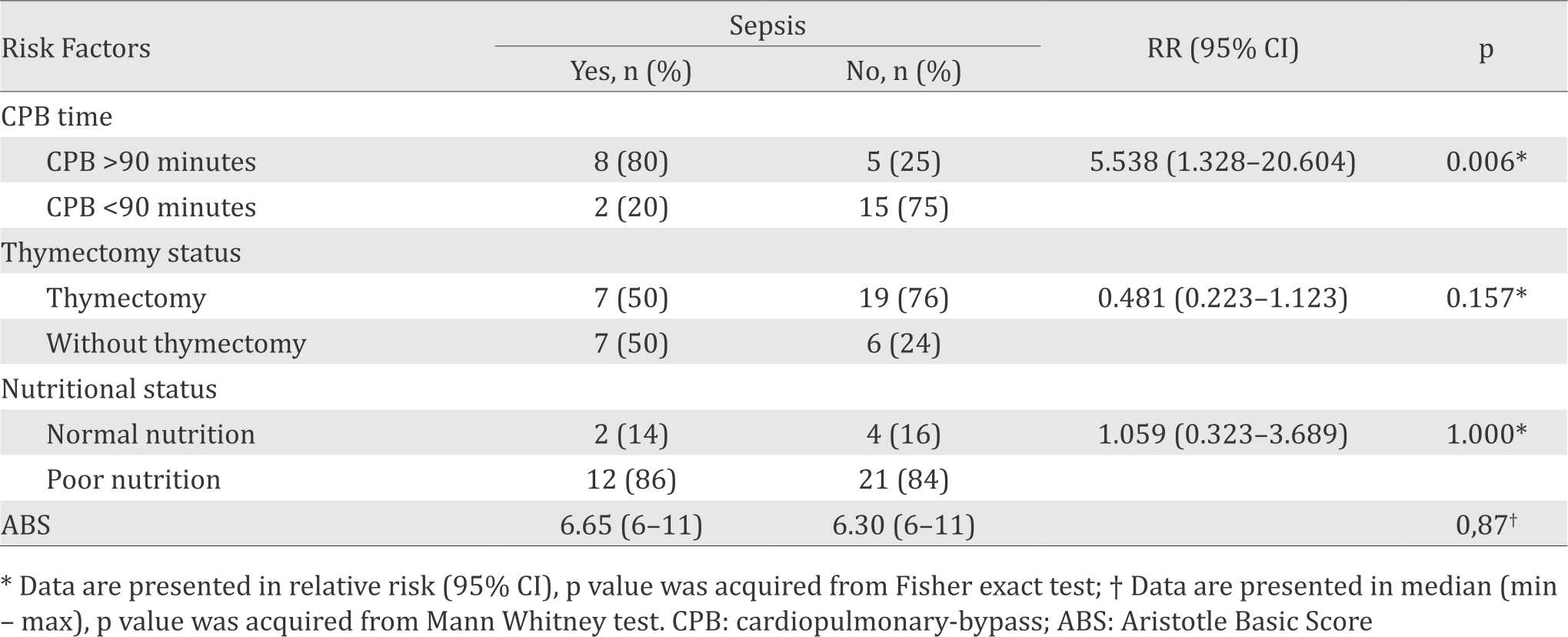
Duration of cardiopulmonary bypass ≥90 minutes was associated with 5.538 increased risk of postsurgical sepsis in comparison to those ≤90 minutes (80% vs 25%, RR=5.538, p=0.006). No association was observed between the incidence of postsurgical sepsis with poor nutritional status (86% vs 84%, RR=1.059, p=1.000), thymectomy (and 50% vs 76%, RR=0.481, p=0.157), and Aristotle Basic Score (p=0.870).
Conclusion
Cardiopulmonary bypass time influences the incidence of sepsis infants undergoing open congenital cardiac surgery. Further studies are needed to elaborate a number of risk factors associated with the incidence of sepsis in this population.
Keywords
aortic cross-clamp, Aristotle Basic Score, cardiopulmonary bypass, congenital heart disease, nutritional status, sepsis
Congenital heart disease (CHD) is a type of heart disorders that occurred before delivery. Malformations in structures of the heart or circulatory functions arise as a result of disruptions, or failures of cardiac development at the early stage of foetal growth.1 In the United States, it is approximated that eight in 1,000 live births suffer from CHD, up to a third of the cases are associated with a complex and fatal cardiac malformation which need urgent surgical correction.2 In Indonesia, according to data from the Indonesian Ministry of Health Centre of Data and Information (Pustadin Kementerian Kesehatan), approximately 36.000 of 4.6 million live births in 2011 were estimated to suffer from CHD. The National Cardiovascular Centre Harapan Kita (NCCHK) could only cover 2.3% of CHD cases in Indonesia annually.
Majority of CHDs are aetiologically unclear. However, gestational diabetes mellitus, infections (i.e. Rubella infection), obesity, smoking, drugs (i.e. retinoic acid, thalidomide, anti-epileptic drugs, antidepressants, and angiotensin-converting enzyme inhibitors), and genetic syndromes (i.e. trisomy 13, trisomy 18, down syndrome, turner syndrome, and other genetic mutations), have been attributed as risk factors.3–5
Congenital heart disease manifests in a variety of signs and symptoms; 2–3 from 1,000 live births in CHD do not show any clinical signs during the first year of life. However, complex cardiac malformation often results in cardiac failure, severe hypoxemia, right or left functional cardiac pumping disruption, and pulmonary hypertension, which increeases the risk of mortality without a prompt surgical correction.3
Corrective surgery is an invasive approach (i.e. open cardiac surgery) which aims to correct functional and anatomical malformation of the heart. Delay in early corrective cardiac surgery has been resulted in a high mortality and morbidity rate.6
A three years study (1991–1994) at the Southampton University Hospital, United Kingdom, demonstrated a 4.4% post cardiac surgery mortality rate (<30 days). This rate was even higher (18%) when the procedure was performed in emergency setting,7 and 29% when performed in a younger age population (i.e. <1 year of age); the study concluded that younger age with low body weight was one of the risk factors associated with high mortality and morbidity rate after cardiac surgery.8–12
In addition to young age, low body weight, and delayed diagnosis and treatment, sepsis has been noted as one of major determinant of the high mortality and morbidity rate after open cardiac surgery. Data from the NCCHK internal database demonstrated a sepsis rate of 15% in patients of older age and low surgical complexity. Further, the overall mortality rate in patients undergoing congenital heart surgery was 8–10%. These numbers may be even higher in children less than one year of age (infants); thus, preventing sepsis will assure a better outcome in these patients.
The present study was conducted to evaluate risk factors [including the effect of cardiopulmonary bypass (CPB) procedure duration (i.e. >90 minutes), nutritional status, surgical complexity as assessed with the Aristotle Basic Score (ABS), and thymectomy associated with the incidence of sepsis after surgery in infants with ABS of six.
METHODS
Forty patients with CHD, aged less than one year old, and ABS ≥6 were observed prospectively for the incidence of sepsis after open cardiac surgery at the Paediatric Congenital Cardiac Surgery Division, NCCHK, were included in the study. Those with prior history of open cardiac surgery, suffered from any associated genetic syndrome and human immunodeficiendy virus (HIV), confirmed with clinical and laboratory assessments, were excluded. Subjects with thymus aplasia, died on surgery, or died less than three days after the surgery were dropped out. Clinical and laboratory analyses were recorded starting at 0 hour (i.e. at the initiation of cardiac surgery) until 192 hours after the surgery. Demographic data including sex, age, weight, height, and nutritional status according to the World Health Organization-National Center for Health Statistics (WHO-NCHS) Z score (age for weight calculation) and clinical data such as blood pressure, heart rate, respiratory rate, temperature, ABS, CPB duration and aortic cross-clamp duration, intubation duration, length of intensive care unit (ICU) stay, and sepsis were recorded. Blood samples were drawn to evaluate serum leukocytes, platelet, C-reactive protein (CRP), lactate, ureum, and creatinine concentration. Congenital cardiac surgical complexity was evaluated according to the ABS.
Diagnosis of sepsis was confirmed according to the surviving sepsis campaign criteria; however, a number of changes in the criteria were made as the study population including children of less than one year of age. Sepsis was confirmed when two or more systemic inflammatory response syndrome signs and symptoms are present: such as rectal temperature >38.3°C or <36°C, tachycardia (heart rate >189 beat/min) or bradycardia (heart rate <90 beat/min), tachypnoea (respiratory rate >34x/min), leukocytosis >17,500/μL or leukopenia <5,000/μL, CRP >10mg/L, procalcitonin ≥2μg/L, vasopressors (epinephrine or norepinephrine in dose of more than 0.05 μg) with any suspicion of infections confirmed by blood or sputum culture on the third day (>72 hours) after surgery.
Prolonged utilization of CPB and aortic crossclamp were defined as CPB time of more than 90 minutes and aortic cross-clamp duration of more than 60 minutes. Mechanical ventilation use of more than 24 hours and ICU stay of more than three days were defined as prolonged intubation time and prolonged ICU stay, respectively.
All numerical data were evaluated for its normality by using the Saphiro-Wilk test. Normally distributed data were presented in mean (standard deviation) while median (minimummaximum) were used when data are not normally distributed. Categorical data were presented in frequency and percentage. Bivariate analyses were conducted using Mann-Whitney, Chi-square, and Fisher Exact test. Logistic regression analysis was performed for multivariate evaluation. All data were assessed using SPSS version 20.
The protocol of this study has been approved by the ethics committee of the National Cardiac Centre, Harapan Kita Hospital, Jakarta (No: LB.05.01.1.4/13/2013).
RESULTS
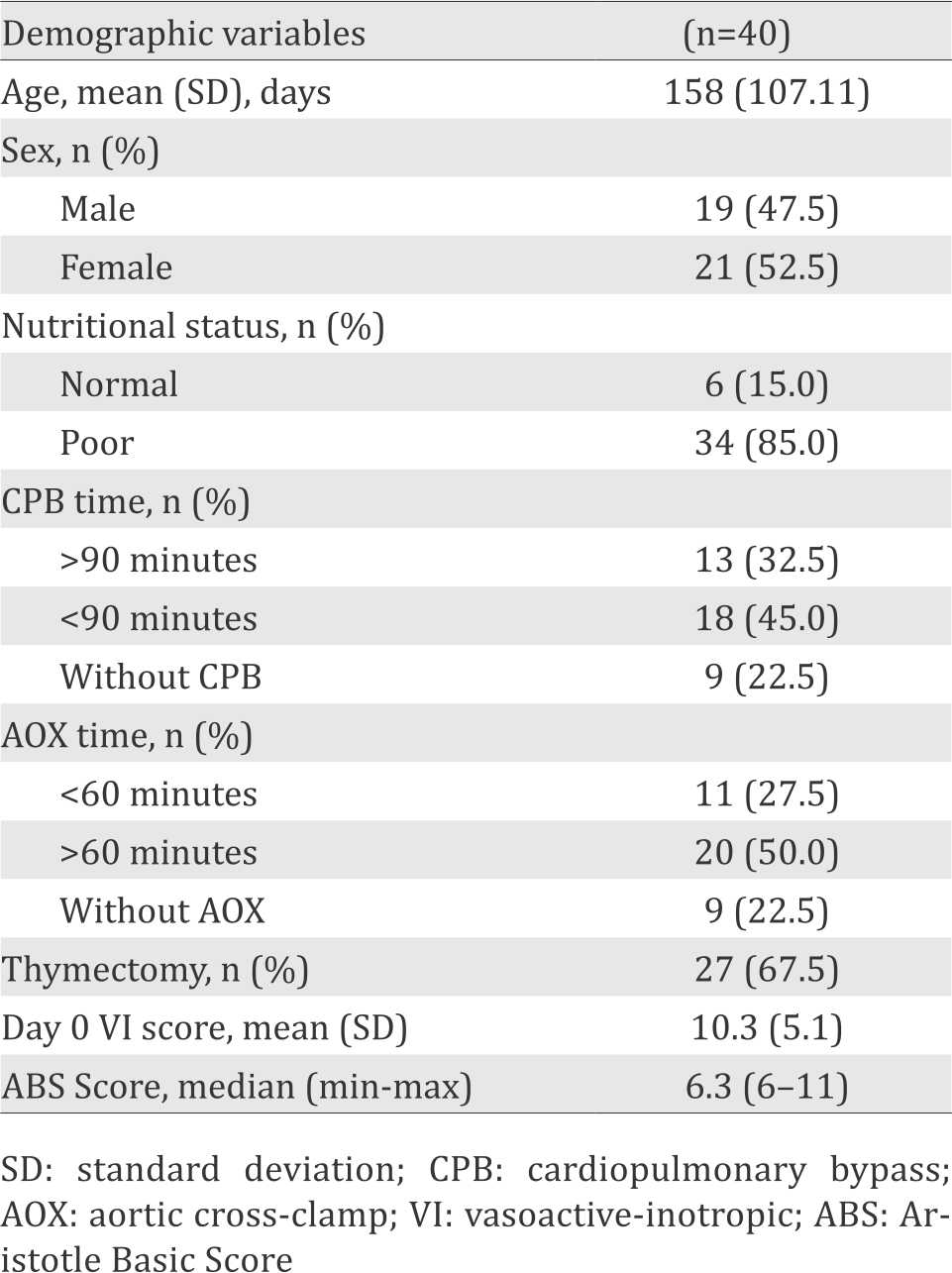
A total of 40 subjects, 19 (47.5%) male and 21 (52.2%) female, were analyzed. Majority of the subjects suffered from a poor nutritional status (85%); the mean age and median ABS were 158 days and 7.355 respectively (Table 1).
Most of the subjects underwent open cardiac surgery with a median CPB duration of 107 minutes; 13 subjects (32.5%) utilized CPB for more than 90 minutes. The median aortic cross-clamp time was 62 minutes; 20 subjects (50%) experienced aortic cross-clamp time of more than 60 minutes. CPB and aortic cross-clamp procedures were not performed on 22.5% of the subjects. Thymectomy was conducted on 27 subjects (67.5%) (Table 1).
Table 1. Demographic and surgical characteristics
Twenty four subjects (60%) were intubated for more than 24 hours, 25 (62.5%) were treated at the intensive care unit for more than 72 hours. Mortality rate and sepsis were 2.5% and 35%, respectively (Table 2). Surgical diagnosis and procedures are presented in Table 3.
Table 2. Postsurgical parameters
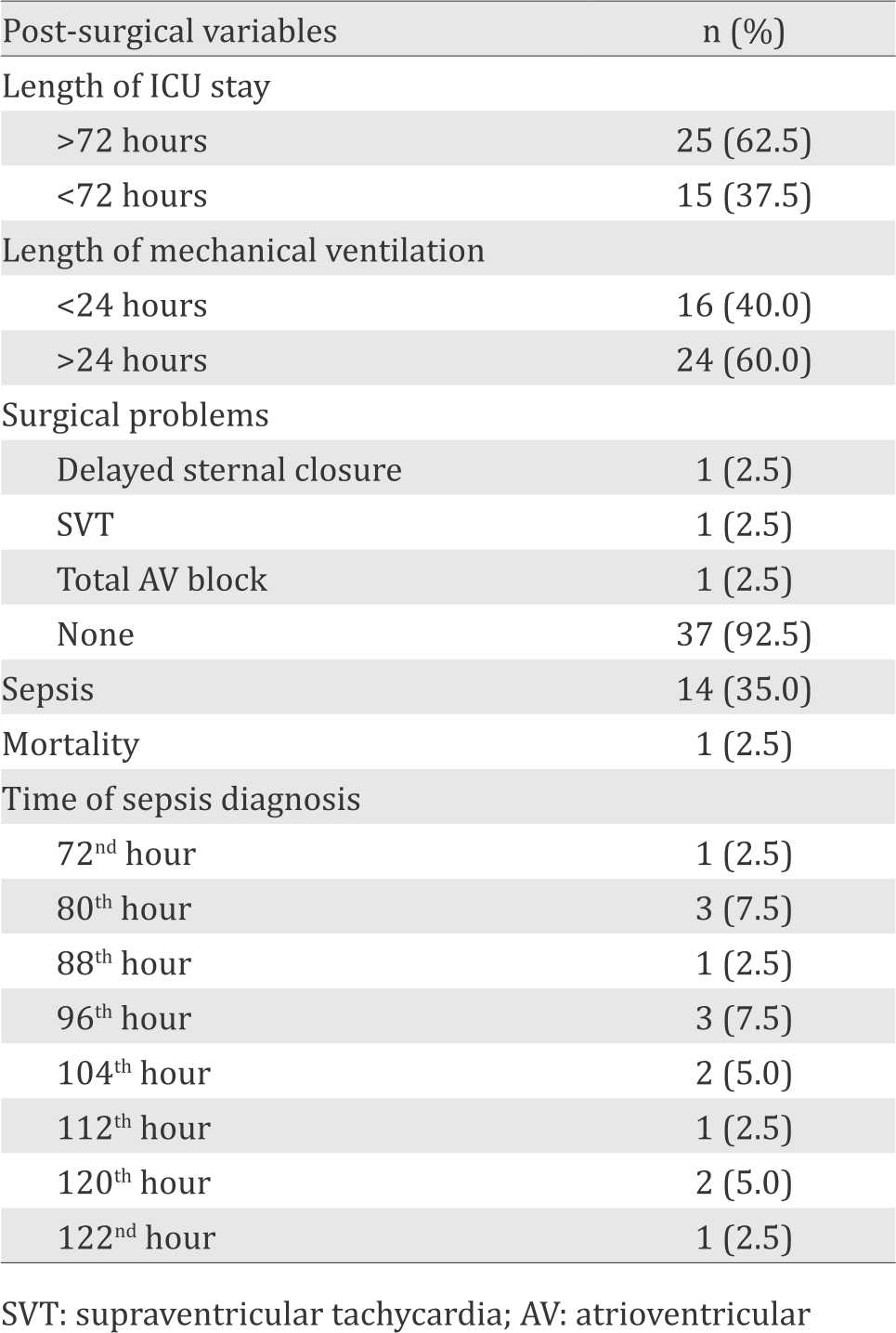
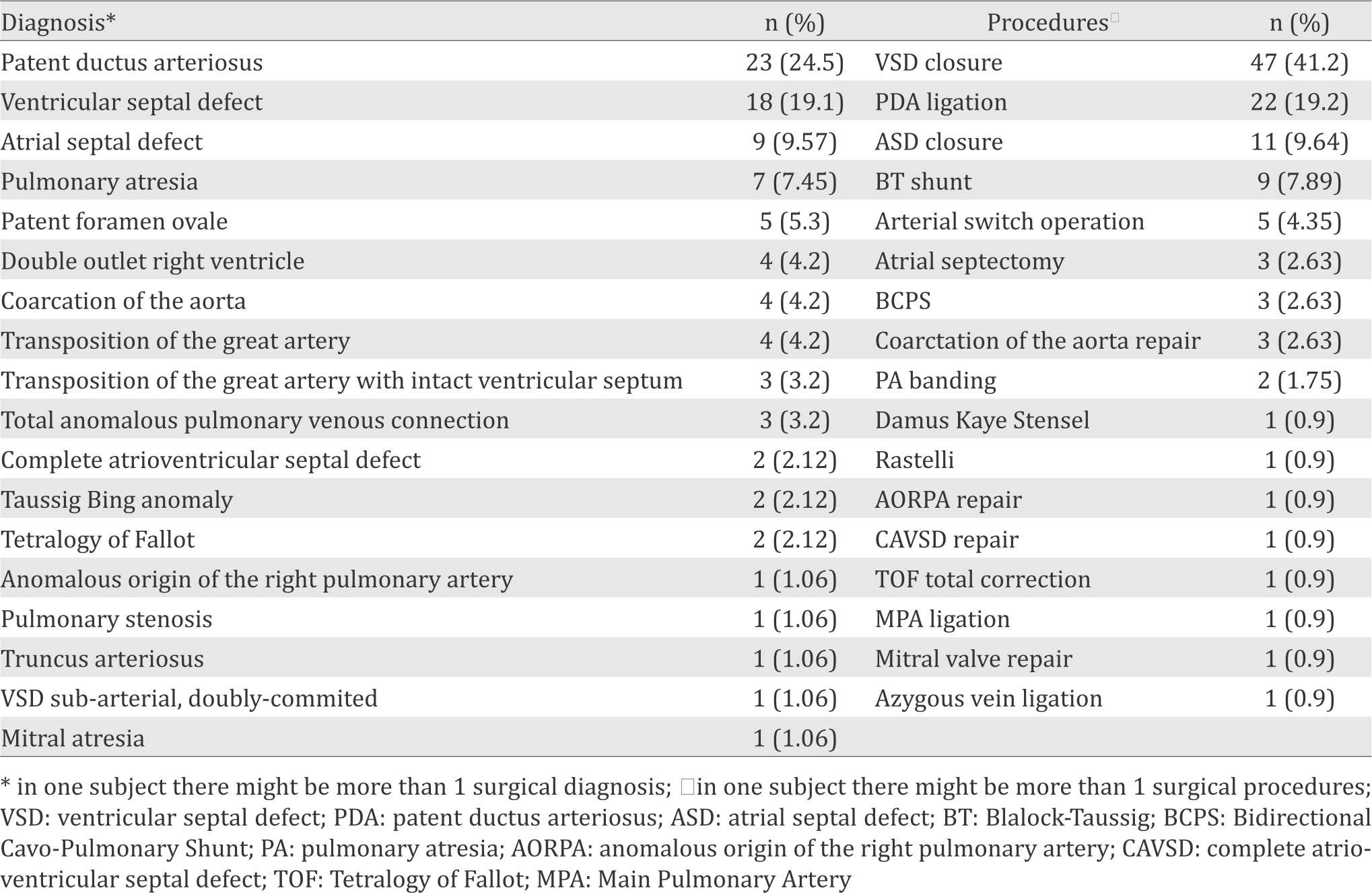
Table 3. Surgical diagnosis and procedures
On Fisher Exact test, subjects who were on CPB for more than 90 minutes were 5.538 more likely to experience sepsis compared to those on CPB for less than 90 minutes (80% vs 20%, RR=5.538, p=0.006). Poor nutritional status was insignificantly associated with 1.059 higher risk of sepsis than normal nutrition (85.7% vs 14.3%, RR=1.059, p=1.000). Similarly, sepsis was observed with a risk of 0.481 times in those who underwent thymectomy than the counterparts (50% vs 50%, RR=0.481, p=0.157); the result, however is not statistically significant. In the study population, there was no statistical significance in the association of ABS and the incidence of sepsis (p=0.870) (Table 4).
Table 4. Bivariate analysis of the risk factors of sepsis
Multivariate logistic regression test was performed to analyze the risk factors of sepsis. The analysis concluded that the risk factors were associated with the incidence of sepsis with R2 of 0.255 and p value of 0.065.
DISCUSSION
Infection is one of the most common problems which occurs after open cardiac surgery. A study by Barker et al13 and colleagues concluded that small baby, complex heart disease, open cardiac surgery, preoperative treatment of more than one day, mechanical ventilation use before surgery, and genetic disorder were closely associated with major postsurgical infections such as mediastinitis, sepsis, and endocarditis.13 Our study analyzed patients who had a high risk of major postsurgical infection (i.e. sepsis) as we included infants with complex cardiac disease and underwent open cardiac surgery.
Sepsis has been a major complication of open cardiac surgery with a high rate of morbidity and mortality.14,15 A number of studies have been conducted to evaluate the outcome of open cardiac surgery in paediatrics however study concerning the incidence of postsurgical sepsis particularly in infants with ABS ≥6 has been limited.
Our pilot study at the NCCHK observed a high rate of sepsis (35%) in infants who underwent open cardiac surgery with ABS ≥6. Postsurgical sepsis has been postulated to be resulted from an imbalance of pro- and anti-inflammatory cytokines release. This inequity further causes a reduction of inflammatory response towards infections. A study by Franke A et al16 and colleagues demonstrated a biphasic curve of immune response induced by open cardiac surgery. On day one (phase one) of the immune response, the pro-inflammatory and anti-inflammatory reactions were originated from the body innate immune response. Balance of proand anti-inflammatory cytokine release on the first phase of immune response occurred on the third day. From the third until the fifth day after the surgery (phase two), adaptive immunity started to react.16 This response was characterized by the release of anti-inflammatory cytokines. These phenomena serve as the basis of early systemic inflammatory response syndrome after open cardiac surgery and explain the reason of sepsis to arise starting from the third day after the surgery. In our study, all incidence of sepsis was diagnosed on day three (the 72nd hour) until day five (the 122nd hour) after surgery (Table 2).
Cardiopulmonary bypass use in open cardiac surgery induces endotoxemia, hemodilution, and systemic inflammation resulted by direct contact activation, ischemic reperfusion injury, and splanchnic hypoperfusion. infants have been the most vulnerable population to suffer from the reactions as their body immune response is immature. This immaturity of immune system causes an increased risk of sepsis and poor outcome after open congenital cardiac surgery compared to the older aged children.17–21
Mortality and mortality after open cardiac surgery were closely associated with CPB duration.22 This result is supported by a study by Chai et al23 and associates who demonstrated that patients who underwent CPB for more than 90 minutes resulted in a more severe pulmonary injury compared to those who were less than 90 minutes in CPB. Additionally, a longer CPB time disrupts the normal functioning of kidney, nervous system, and gastrointestinal tract; thus, increases morbidity and mortality after surgery.23–29 Gastrointestinal tract vascular integrity and perfusion were disrupted (i.e. sphlanchnic hypoperfusion) by a long CPB time (>90 minutes). Further, cardiac malfunctioning alone might cause damages in the alimentary tract mucosal barrier resulting in bacterial and endotoxins translocation to systemic circulation and sepsis. In accordance to this hypothesis, Klein et al30 stated that the duration of cardiopulmonary bypass increased the risk of endotoxemia and postsurgical infection.30 Further, complex congenital cardiac diseases were substantially related with the risk of necrotizing enterocolitis. Even though the enterocolitis was not directly associated with mortality rate, 20% of patients with this inflammatory reaction would die.31
In our study, major infection (i.e. sepsis) was observed to be significantly higher in patients with CPB duration of more than 90 minutes compared to those with shorter CPB duration (80% vs 20%, RR=5.538, p=0.005). Inferior postsurgical outcomes were also observed in this study group with prolonged ICU stay (64.6% vs 35.3%, p=0.005) and prolonged mechanical ventilation (68.8% vs 31.2%, p=0.002). These results are in accordance with the previous trials which demonstrated that CPB time was associated with the incidence of sepsis, morbidity, and mortality after open congenital cardiac surgery particularly in younger aged children with complex cardiac malformation. Endotoxemia and necrotising enterocolitis (NEC) might also play a role in the high incidence of sepsis in our study population; unfortunately, we did not elaborate it further.
Thymectomy has been known to influence the population of T lymphocytes, serum CD8 and CD4 concentration after surgery.32 Decreased in number and function of T lymphocytes has been postulated to be one of the causes of sepsis after open cardiac surgery with thymectomy.33 Our study, on the contrary, did not observe a significant relation between sepsis rate and thymectomy (RR=0.481, p=0.157). A number of previous studies have been conducted to evaluate the association of thymectomy and outcomes of open cardiac surgery with partial, total, or without thymectomy and resulted in similar answers. There was one study, however, demonstrating a significantly lower thymic recent emigrant cells and lymphocytes subpopulation in patients who were thymectomized compared to the counterparts. The incidence of postsurgical immunodeficiency, recurrent infections, neoplasm, or autoimmune reactions was also not significantly related with thymectomy procedures.32,34,35 This occurrence might be explain by an incomplete thymectomy, release of mature T cells before the procedure, compensatory increase in the nonspecific immune responses such as neutrophils and natural killer cells, and activation of other reticuloendothelial cells.34
Failure to thrive and poor nutritional status have been considered as one of the major problems in children with congenital cardiac anomaly. Up to 40% of children with congenital heart problem suffered from failure to thrive and a poor nutritional status. Nutritional status is closely associated with immune response and outcomes of open cardiac surgery.36–38 A study in paediatric population in Bangladesh demonstrated that a poor birth weight was associated with immunity disruptions manifested in a low CD3 concentration, accelerated peripheral T cells death, and increased turn over cycle of T cells by thymus. All of the processes resulted in decreased immunity towards infection in children. Additionally, a low birth weight increases the risk of infections in later life.39 A decrease in immune response resulted from poor nutritional status has been one of the most important risk factors of alteration in inflammatory responses after open cardiac surgery. Hassen et al40 observed an increased risk of systemic inflammatory response syndrome after a major blood vessel surgery in patients with protein energy deficiency.40 Further, in a population of children after Fontan surgery demonstrated that Z-score of less than -2 was significantly associated with mortality, surgical failure, and length of hospital stay.41 Nutritional status calculated by weight for age formula and young age were correlated with length of stay and hemodynamic status after a Glenn bidirectional surgery.42 While accompanied with hypoalbuminemia, a poor nutrition in children with congenital heart disease increased the risk of mortality and postsurgical infections as well as prolonged hospital stay.43
In our observation, 85.7% of the subjects who suffered from sepsis had a poor nutritional status; this finding, however, showed no significant finding on bivariate analyses (RR=1.059, p=1.000). Our finding is supported by previous study by Azakie et al44 in which they concluded that low birth weight or prematurity was related with a poor outcome in children who underwent open cardiac surgery; yet, the finding was not statistically significant.44 These data might imply a better development of open cardiac surgical approach in premature or low and very low birth weight baby; hence, the previously significantly related risk factors were no longer found to be essential today.
Aristotle basic score (ABS) is one approach to evaluate complexity in congenital cardiac surgery. ABS consists of three main components: (1) mortality, (2) morbidity (i.e. length of ICU stay), and (3) surgical difficulty.45,46 Subjects with sepsis in our study had a median ABS of 6.65 (6–11). There was no statistically significant correlation between ABS and sepsis rate. This insignificant finding might be resulted from the fact that ABS is the simplest method to evaluate and project complexity and outcomes after congenital cardiac surgery without considering patients characteristics such as anatomical variations, associated surgical procedures, age, and co-morbidities in the assessment as the Aristotle Comprehensive Score (ACS). A study by Kansy and colleagues analyzed the outcome of open congenital cardiac surgery with surgical complexity evaluated with four different stratification methods: (1) Aristotle Basic Score; (2) Risk Adjustment for Congenital Heart Surgery-1 (RAHCS-1 Categories); (3) The Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery Congenital Heart Surgery Mortality Score (STAT Mortality Score); and (4) STAT Mortality Categories.47 The study observed that STAT Mortality Score had a stronger correlation with mortality outcome. Insignificant result between ABS and sepsis in our analysis might also be resulted from a better management of patients with a complex congenital cardiac disease which in turn significantly reduced the rate of morbidity and mortality after surgery.
In conclusion, the risk factors evaluated in our study could only explain 25.5% variability of sepsis. CPB time of more than 90 minutes was significantly related with sepsis rate after open cardiac surgery in infants with ABS ≥6. Further study is needed to elaborate the role of a number of risk factors associated with sepsis in this population.
Conflicts of Interest
The authors affirm no conflict of interest in this study.
Acknowledgment
None.
REFERENCES
- Allen HD, Driscoll DJ, Shaddy RE, Feltes TF. Moss and Adams’ heart disease in infants, children, and adolescents including the fetus and young adult. Philadelphia: Lippincott Williams & Wilkins; 2008. p. 597–639.
- David WB, David RF. Congenital Heart Disease in Children And Adolescents. In: Richard AW, Robert AH, Valentin F, editors. Hurst’s the heart. 13th ed. United States: The McGraw-Hill Companies Inc; 2011.
- Daniel B. Epidemiology and genetic basic of congenital heart disease. In: Kliegman R, Behrman R, Jenson H, editors. Nelson Textbook of Pediatrics. 19th ed. Philadelphia: Saunders Elsevier. 2011.
- Mills JL, Troendle J, Conley MR, Carter T, Druschel CM. Maternal Obesity and Congenital Heart defect: a Population –based Study. Am J Clin Nutr. 2010;91:1543–9.
- Rahayuningsih SE. Detection of NKX2.5, TBX5, GATA4, and MYH6 Gene Mutations in Finding an Association with Sporadic Secundum Atrial Septal Defect. Majalah Kedokteran Indonesia. 2009;59:315–21.
- Wallace MC, Jaggers J, Li JS, Jacobs ML, Jacobs JP, Benjamin DK, et al. Center variation in patient age and weight at Fontan operation and impact on postoperative outcomes. Ann Thorac Surg. 2011;91(5):1445–52.
- Lee AH, Borek BT, Gallagher PJ, Saunders R, Lamb RK, Livesey SA, et al. Prospective study of the value of necropsy examination in early death after cardiac surgery. Heart. 1997;78:34–8.
- Aylin P, Bottle A, Jarman B, Elliott P. Paediatric cardiac surgical mortality in England after Bristol: descriptive analysis of hospital episode statistics 1991-2002. BMJ. 2004;329(7470):825.
- Limperopoulos C, Majnemer A, Shevell MI, Rosenblatt B, Rohlicek C, Tchervenkov C, et al. Functional limitations in young children with congenital heart defects after cardiac surgery. Pediatrics. 2001;108(6):1325–31.
- Catre D, Lopes MF, Madrigal A, Oliveiros B, Viana JS, Cabrita AS. Early mortality after neonatal surgery: analysis of risk factors in an optimized health care system for the surgical newborn. Rev Bras Epidemiol. 2013;16(4):943–52.
- Oppido G, Pace Napoleone C, Formigari R, Gabbieri D, Pacini D, Frascaroli G, et al. Outcome of cardiac surgery in low birth weight and premature infants. Eur J Cardiothorac Surg. 2004;26(1):44–53.
- Kochilas LK, Vinocur JM, Menk JS. Age-dependent sex effects on outcomes after pediatric cardiac surgery. J Am Heart Assoc. 2014:3(1)-e000608.
- Barker GM, O’Brien SM, Welke KF, Jacobs ML, Jacobs JP, Benjamin DK Jr, et al. Major infection after pediatric cardiac surgery: a risk estimation model. Ann Thorac Surg. 2010;89(3):843–50.
- Shane AL, Stoll BJ. Neonatal sepsis: progress towards improved outcomes. J Infect. 2014;68(Suppl1):S24–32.
- Valera M, Scolfaro C, Cappello N, Gramaglia E, Grassitelli S, Abbate MT, et al. Nosocomial infection in pediatric cardiac surgery, Italy. Infect Control Hosp Epidemiol. 2001;22(12):771–5.
- Franke A, Lante W, Fackeldey V, Becker HP, Thode C, Kuhlmann WD, et al. Proinflamatory and antiinflamatory cytokines after cardiac operation: different cellular sources at different times. Ann Thorac Surg. 2002;74(2):363–70.
- Wynn J, Cornell TT, Wong HR, Shanley TP, Wheeler DS. The host response to sepsis and developmental impact. Pediatrics. 2010;125(5):1031–41.
- Larmann J, Theilmeier G. Response to cardiac surgery: cardiopulmonary bypass versus non-cardiopulmonary bypass surgery. Best Prac Res Cl Anaesth. 2004;18(3):425–38.
- Paparella D, Yau TM, Young E. Cardiopulmonary bypass induced inflammation: pathophysiology and treatment. An Update. Eur J Cardiothoracic Surg. 2002;21:232–44.
- Kapoor MC, Ramachandran TR. Inflammatory response to cardiac surery and strategies to overcome it. Ann Card Anaesth. 2004;7(2):113–28.
- Wan S, LeClerc JL, Vincent JL. Inflammatory response to cardiopulmonary bypass: mechanisms involved and possible therapeutic strategies. Chest. 1997;112(3):676–92.
- Salis S, Mazzani VV, Merli G, Salvi L, Tedesco CC, Veglia F, et al. Cardiopulmonary bypass duration is an independent predictor of morbidity and mortality after cardiac surgery. J Cardiothorac Vasc Anesth. 2008;20(6):814–22.
- Chai PJ, Williamson JA, Lodge AJ, Daggett CW, Scarborough JE, Meliones JN, et al. Effect of ischemia on pulmonary dysfunction after cardiopulmonary bypass. Ann Thorac Surg. 1999;67(3):731–5.
- Tsuchida M, Watanabe H, Watanabe T, Hirahara H, Haga M, Ohzeki H, et al. effect of cardiopulmonary bypass on cytokine release and adhesion molecule expression in alveolar macrophage. Preliminary report in six cases. Am J Respir Crit Care Med. 1997;156(3 Pt 1):932–8.
- Gormley SM, McBride WT, Armstrong MA, Young IS, McClean E, MacGowan SW, et al. Plasma and urinary cytokinie homeostasis and renal dysfunction during cardiac surgery. Anesthesiology. 2000;93(5):1210–6.
- Brown WR, Moody DM, Challa VR, Stump DA, Hammon JW. Longer duration of cardiopulmonary bypass is associated with greater number of cerebral microemboli. Stroke. 2000;31(3):707–13.
- Dong GH, Wang CT, Li Y, Xu B, Qian JJ, Wu HW, et al. Cardiopulmonary bypass induced microcirculatory injury of the small bowel in rats. World J Gastroenterol. 2009;15(25):3166–72.
- Jonas RA, Wypij D, Roth SJ, Bellinger DC, Visconti KJ, du Plessis AJ, et al. The influence of hemodilution on outcome after hypothermic cardiopulmonary bypass: result of a randomized trial in infants. J Thorac Cardiovasc Surg. 2003;126(6):1765–74.
- Siddiqui MMA, Paras I, Jalal A. Risk factors of prolonged mechanical ventilation following open heart surgery: what has changed over the last decade? Cardiovasc Diagn Ther. 2012;2(3):192–9.
- Klein DJ, Briet F, Nisenbaum R, Romaschin AD, Mazer CD. Endotoxemia related to cardiopulmonary bypass is associated with increased risk of infection after cardiac surgery: a prospective observational study. Crit Care. 2011;15(1);1–6.
- McElhinney DB, Hendrick HL, Bush DM, Pereira GR, Stafford PW, Gaynor JW, et al. Necrotizing enterocolitis in neonates with congenital heart disease: risk factors and outcomes. Pediatrics. 2000;106(5):1080–7.
- Mancebo E, Clemente J, Sanchez J, Ruiz-Contreras J, De Pablos P, Cortezon S, et al. Longitudinal analysis of immune function in the first 3 years of life in thymectomized neonates during cardiac surgery. Clin Exp Immunol. 2008;154(3):375–83.
- Bayer A, Doğan OF, Ersoy F, Ersoy U. The effect of open heart surgery on circulatin lymphocytes and lymphocyte subsets in pediatrics patients. Turk J Thorac Cardiovasc Surg. 2009:17(1):13–17.
- Eysteindottir JH, Freysdottir J, Haraldsson A, Stefansdottir J, Skaftadottir R, Helgason H, et al. The influence of partial or total thymectomy during open heart surgery in infants on the immune function later in life. Clin Exp Immunol. 2004;136(2):349–55.
- Madhok AB, Chandrasekran A, Parnell V, Gandhi M, Chowdhury D, Pahwa S. Level of recent thymic emigrant cells decrease in children undergoing partial thymectomy during cardiac surgery. 2005;12(5):563–5.
- Cameron JW, Rosenthal A, Olson AD. Malnutrition in hospitalized children with congenital heart disease. Arch Pediatr Adolesc Med. 1995;149(10):1098–102.
- Katona P, Katona-Apte J. The interaction between nutrition and infection. Clin Infect Dis. 2008;46(10):1582–8.
- Calder PC, Jackson AA. Undernutrition, infection and immune function. Nutr Res Rev. 2000;13(1):3–29.
- Raqib R, Alam DS, Sarker P, Ahmad SM, Ara G, Yunus M, et al. Low birth weight is associated with altered immune function in rural banglades children: a birth cohort study. Am J Clin Nutr. 2007;85(3):845–52.
- Hassen TA, Pearson S, Cowled PA, Fritdge RA. Preoperative nutritional status predict the severity of the systemic inflammatory response syndrome (sirs) following major vascular surgery. Eur J Vasc Endovasc Surg. 2007;33:696–702.
- Wallace MC, Jaggers J, Li JS, Jacobs ML, Jacobs JP, Benjamin DK, et al. Center variation in patient age and weight at Fontan operation and impact on post-operative outcomes. Ann Thorac Surg. 2011;91(5):1145–52.
- Anderson JB, Beekman RH, Border WL, Kalkwarf HJ, Khoury PR, Uzark K, et al. Lower weight-for-age z score adversely affects hospital length of stay after the bidirectional Glenn procedure in 100 infants with a single ventricle. J Thorac Cardiovasc Surg. 2009;138(2):397–404.
- Leite HP, Fisberg M, de Carvalho WB, de Camargo Carvalho AC. Serum albumin and clinical outcome in pediatric cardiac surgery. Nutrition. 2005;21(5):553–8.
- Azakie A, Johnson NC, Anagnostopoulos PV, Egrie GD, Lavrsen MJ, Sapru A. Cardiac surgery in low birth weight infants: current outcomes. J Cardiovasc Thorac Surg. 2012;12(3):409–14.
- Lacour-Gayet F, Clarke DR, Aristotle Committee. The Aristotle method: a new concept to evaluate quality of care based on complexity. Curr Opin Pediatr. 2005;17(3):412–7.
- Lacour-Gayet F, Clarke D, Jacobs J, Comas J, Daebritz S, Daenen W, et al. The Aristotle score: a complexityadjusted method to evaluate surgical result. Eur J Cardiothorac Surg. 2004;25(6):911–24.
- Kansy A, Maruszewski B, Jacobs J, Maruszewski P. Application of four complexity stratification tools (Aristotle Basic Score, RACHS-1, STAT Mortality Scorem and STAT Mortality Categories) to evaluate early congenital heart surgery outcomes over 16 years at a single institution. Kardiochir Thorakochir Polsk. 2013;10(2):115–9.
Copyright @ 2016 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id