
Section Abstract Introduction Methods Results Discussion Conflict Of Interest Acknowledgment References
Clinical Research
Serum specific IgE responses to inhalant allergens sensitization
pISSN: 0853-1773 • eISSN: 2252-8083
http://dx.doi.org/10.13181/mji.v26i3.1909 Med J Indones. 2017;26:224–8
Received: March 16, 2017
Accepted: September 06, 2017
Author affiliation:
1 Department of Internal Medicine, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia
2 Department of Internal Medicine, Faculty of Medicine, Universitas Hasanuddin, Makassar, Indonesia
3 Department of Internal Medicine, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
Corresponding author:
Iris Rengganis
E-mail: irisrengganis@yahoo.com
Background
Serum specific immunoglobulin E (ssIgE) sensitization to common inhalant allergens has not been studied in Indonesia. This study aimed to evaluate specific IgE production of common inhalant allergens in patients with asthma and/or allergic rhinitis in Jakarta, Indonesia.
Methods
This was a cross-sectional study in adult patients with respiratory allergy from September to December 2016 at Cipto Mangunkusumo Hospital, Jakarta. Patients were included if they showed at least one positive skin prick test (SPT) to environmental allergens. Serum specific IgE was assayed by using multiple allergosorbent methods. Inhalant allergens tested were dust mites, pollen, cockroach, animal dander, and mould. Serum IgE level more than 0.35 kU/L was considered positive.
Results
One hundred subjects were enrolled (76% women). Dust mites made up 75% of sensitization, followed by cat/ dog (31%), cockroach (27%), pollen (24%), and mould (6%). Almost all patients sensitized to cockroach, pollen, cat/dog epithelia and mould were also co-sensitized with dust mites. Twenty two percent of patients were negative to all tested allergens.
Conclusion
IgE-sensitization to inhalant allergens varies widely in respiratory allergic patients. House dust and storage mites are the most common allergens. About onefifth of the subjects did not show specific-IgE sensitization. Thus, this test should always be combined with SPT to diagnose allergy.
Keywords
allergic rhinitis, asthma, house dust mites, IgE sensitization, specific IgE
Respiratory allergic disease is often diagnosed based on the patient’s history and experience. Sometimes it is difficult to associate symptoms with a specific allergen exposure, and patient subjective impression to an allergen trigger can also be misleading.1 An accurate and comprehensive identification of inhalant allergen sensitization is important to plan allergen avoidance and immunotherapy. Skin prick test (SPT) is currently the gold standard to evaluate allergic sensitization. It is usually performed on the volar aspect of the forearm, which can accommodate 20– 26 allergens simultaneously. A positive SPT result means that the person is sensitive or has been sensitized to the tested allergen.2 At molecular level, it shows the propensity of an individual to develop immunoglobulin E (IgE) antibodies against common environmental allergens and not necessarily being allergic to the allergen. Previous study using SPT in Jakarta showed that respiratory allergies were usually caused by environmental inhalant allergens such as dust mites (Dermatophagoides pteronyssinus, Dermatophagoides farinae, and Blomia tropicalis), German cockroach (Blatella germanica), tree and grass pollens.3–5 However, although considered safe and inexpensive, SPT cannot be performed in patients under antihistamines or corticosteroid treatment and those who have serious skin disorder.
Detection of serum specific IgE (ssIgE) is another method to assess allergic sensitization. Serum collection is more convenient to the patient and may allow detection of more than 25 specific allergens from a single blood collection. Although imperfect, several preliminary studies have shown good agreement between ssIgE and SPT.6 Serum specific IgE testing is now accepted as a part of diagnostic work-up for allergy diseases.7 This test has long been available in Indonesia for diagnosing allergy diseases such as respiratory and skin allergy. However, the previously available panel tests were not specifically made based on local allergens of Indonesia. Based on our proposal, a newly designed IgE-specific panel test consisting of 44 and 54 local allergens is now available with the assistance of two European companies. As previous studies using SPT have shown that respiratory allergic patients are commonly sensitized to inhallant allergen,3–5 we conducted this study to evaluate the specific IgE production to common inhalant allergens in patients with asthma and/or allergic rhinitis in Jakarta, Indonesia.
METHODS
Study design and subjects
This was a cross-sectional study in patients with a history of respiratory allergy in Jakarta, Indonesia between September and December 2016. Adult asthmatic and/or allergic rhinitis patients aged 19–60 years were invited to undergo serum specific IgE testing at the Allergy and Immunology Clinic, Cipto Mangunkusumo Hospital, Jakarta. Patients were included if they showed at least one positive skin prick test with main environmental allergens causing allergy in Indonesia. Patients gave a written informed consent prior to enrollment of this study. Ethical approval was granted by the Ethical Committee of Medical Research, Faculty of Medicine, University of Indonesia No. 796/UN2.F1/ ETIK/2016.
Specific IgE Measurement
Quantitative determination of specific IgE in serum was carried out by immunoblot method known as multiple allergosorbent (MAST) assays. In order to give more comprehensive data, we used allergen panel tests provided by Euroline® (Euroimmun, Germany) and Alleisa Screen® (Mediwiss, Germany) as per proposal. The allergens include: House dust mite (D. pteronyssinus, D. farinae, D. microceras, B. tropicalis, T. putrescentiae, G. domesticus, and A. siro), Pollen allergens (Bermuda grass, timothy grass, grass mixed, acacia pollen, australian pine, pine mixed, maize, and oil palm), Cockroach allergens (German cockroach and cockroach mixed) and Animal dander allergens (cat and dog) and mould allergen (Mould mix).
Standards of specific IgE concentrations were ranged from 0.35 to 100 kU/L. Both manufactures defined a positive result as serum sIgE level more than 0.35 kU/L. The results were also reported as categories as follows: Class 0: <0.35 kU/L no specific antibody detection. Class 1:0.35–0.7 kU/L very weak antibody concentration. Class 2:0.70–3.49 kU/L weak antibody concentration. Class 3:3.50–17.49 kU/L clear antibody concentration. Class 4:17.50–49.99 kU/L strong antibody concentration. Class 5:50.00–100.00 ku/L very strong antibody concentration. Class 6:>100.00 kU/L extremely high antibody concentration.
RESULTS
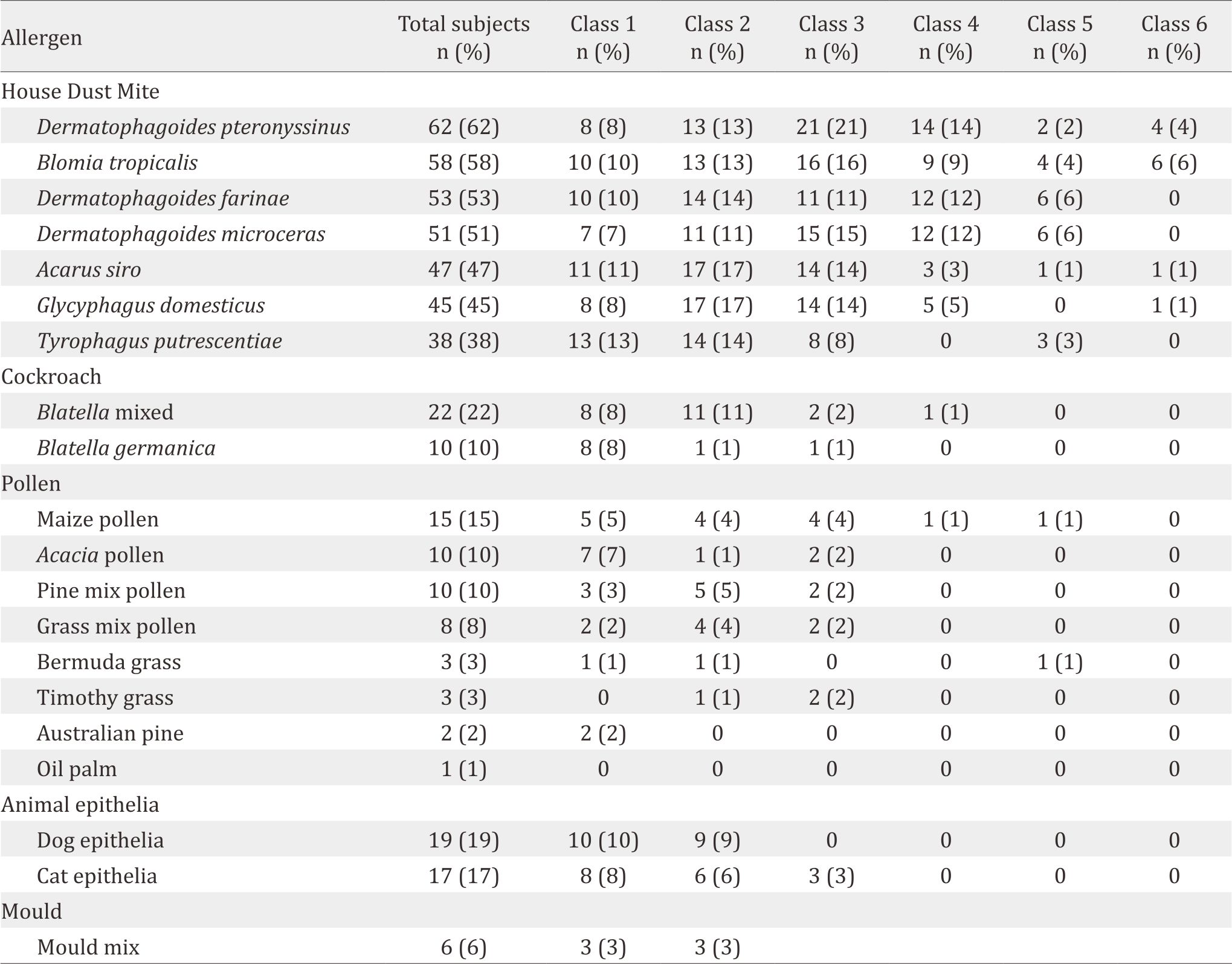
Characteristics of the study subjects are shown in Table 1. and distribution of positive result in number of patients to specific IgE sensitization (n=100) is shown in Table 2. The most common allergen were D. pteronyssinus. B. tropicalis, D. farinae, and D. microceras were also important house dust allergen in Indonesia (more than 50% sensitization). It means that these allergens should also be avoided by allergic patients. The sensitization of cockroach, pollen, animal epithelia and mould were very low.
Table 1. Characteristics of the study subjects (n=100)
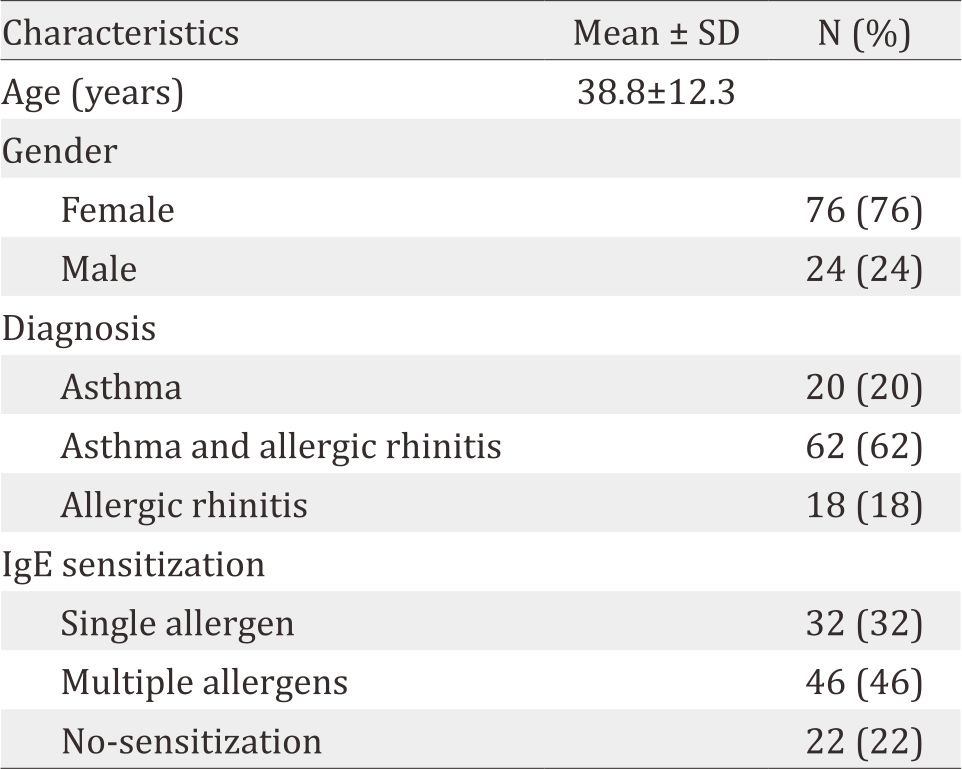
Table 2. Distribution of specific IgE sensitization (n=100)
DISCUSSION
This is the first study to assess the IgEsensitization among adult respiratory allergic patients in Indonesia. Serum-specific IgE testing only cannot be used to establish the diagnosis of allergic disease. Other diagnostic measures are needed. In our study, 22% of patients did not show sensitization to any tested allergen although subjects’ inclusion criteria has included at least one positive SPT. This could be due to the fact that ssIgE has lower sensitivity than SPT to diagnose allergy disease. Discordance between ssIgE and SPT results is common; therefore, it is advisable to use both methods complimentarily.1
House dust mites showed the highest prevalence of sensitization, which were predominated by three species, i.e. D. pteronyssinus, D. farinae, and B. tropicalis. These 3 species were accounted for 73% of all IgE-sensitization or 97.3% of mite-IgEsensitizations in our study. It was not surprising because house dust mites has been the major source of allergens in people throughout the world.8 There are more than 250 dust mite species causing health problems, but only the pyroglyphid mites (D. pteronyssinus and D. farinae) that are responsible for more than 90% of house dust mites allergies worldwide.9 D. microceras, is the third Dermatophagoides found in this study and could be an important house dust mites allergen. A study in Taiwan found that IgE-sensitization to D. microceras among 579 asthmatic patients was quite high (59%), almost similar to D. pteronyssinus (59.8%) and slightly higher than D. farinae (56.8%).10–11 B. tropicalis is a storage mite which its importance is getting higher as inhalant house dust allergen in urban environment in Asia.12 Another study in Taiwan reported that 40% sensitization to B. tropicalis was among asthmatic patients.13 Another Taiwanese study on asthmatic children aged 3–15 years found that sensitization to D. microceras was in 84%, not too much different from D. pteronyssinus (87%) and D. farinae (85%), but it was higher than B. tropicalis (65%).14
A. siro, T. putrescentiae, and G. domesticus are not very important in Indonesia. G. domesticus is also known as furniture mite and is found in foods, grains in warehouses, and other storage areas.15 This mite may also induce asthma and rhino-conjunctivitis symptoms in sensitized individuals.16 T. putrescentiae and A. siro are other storage mites that belong to the family of Acaridae. T. putrescentiae lives in hot, humid climates and is a common pest of stored food products.17 It was reported to be a dominant species of storage mites in Korea and caused IgE reactivity.18
Strong association between sensitization status and total IgE values, and the striking co-sensitization among biologically unrelated allergens suggest that polysensitization is the expression of a distinct clinical, more severe, atopic phenotype, rather than of biological crossreactivity to similar allergens.19 In this study, we found that 48 patients were sensitized to both B. tropicalis and D. pteronyssinus allergens. Concurrent sensitization of both species was also reported in 63.3% of asthmatic patients in Taiwan.13 Further study confirmed that about 18% of patients sensitized to B. tropicalis might have been caused by a cross-reaction.20
In addition to dust mites, our study also found sensitization to cat/dog dander (31%), cockroach (27%), and pollen (24%). Mould might not have clinical significance since only a small number of patients were sensitized to it. Pet dander and cockroach are also important allergens in Asia. A study in Taiwan showed that IgE sensitization to cockroach was 38.3% whereas sensitization to dog dander was found in 26.3%; cat dander in 10%, Candida albicans in 13.3%, and Cladosporium herbarum in 6.6%.10
Serum specific IgE is superior compared to SPT in testing more variant of allergens in one time. However, in this descriptive study, we can only measure the degree of sensitization based on level of ssIgE positivity. To assess whether this ssIgE has a correlation to the clinical manifestation of the patient, a further diagnostic study is needed. In conclusion, IgE-sensitization to inhalant allergens varies widely in respiratory allergic patients. House dust and storage mites comprises 75% of all sensitization to the tested allergen. Co-sensitization among inhalant allergens is also common, and 80% occurs with dust mites. However, 22% of the subjects failed to show IgE sensitization, confirming the importance combining serum IgE test with skin prick test to diagnose allergy. Sensitization to D. microseras was quite high in our study. Thus, this allergen can be considered to be included in standard allergen of SPT in Indonesia.
Conflicts of Interest
All of the authors have no relevant financial affiliations to disclose. This study was partly supported by Euroline®, Euroline® (Euroimmune) and Alleisa Screen® (Mediwiss Companies), which providing the reagents for this study
Acknowledgment
None.
REFERENCES
- De Vos G. Skin testing versus serum-specific IgE testing: which is better for diagnosing aeroallergen sensitization and predicting clinical allergy? Curr Allergy Asthma Rep. 2014;14:430.
- Pawankar R, Canonica GW, Holgate ST, Lockey RF, Blaiss M. The world allergy organization (WAO) white book on allergy: Update 2013;WAO:87–92.
- Baratawidjaja IR, Baratawidjaja PP, Darwis A, Hwe LS, Tim CF, Wah LB, et al. Prevalence of allergic sensitization to regional inhalant among allergic patient in Jakarta, Indonesia. Asian Pac J Allergy Immunol.1999;17:9–12.
- Sundaru H. House dust mite allergen level and allergen sensitization as risk factor for asthma among student in Central Jakarta. Med J Indones. 2006;15:55–9.
- Rengganis I, Hartana A, Guhardja E, Djauzi S, Budiarti S. Pollen sensitivity among respiratory allergic patients. Majalah Kedokteran Indonesia. 2008;58:327–34. Indonesian.
- Calabria CW, Dietrich J, Hagan L. Comparison of serumspecific IgE (ImunoCAP) and skin prick test result for 53 inhalant allergens in patients with chronic rhinitis. Allergy Asthma Proc. 2009;3:386–90.
- Canonica GW, Ansotegui IJ, Pawankar R, Schmid- Grendelmeier P, van Hage M, Baena-Cagnani CE, et al. A WAO – ARIA – GA2LEN consensus document on molecular-based allergy diagnostics. WAO J. 2013;6:17.
- Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A et al. Allergic rhinitis and its impact on asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allergy 2008;63 (Suppl 86):8–160.
- Thomas WR. Geography of house dust mite allergens. Asian Pac J Allergy Immunol. 2010;28:211–24.
- Sidenius KE, Hallas TE, Poulsen LK, Mosbech H. House dust mites and their allergens in Danish mattresses – results from a p.
- Chiang CH, Wu KM, Wu CP, Yan HC, Perng WC. Evaluation of risk factors for asthma in Taipei City. J Chin Med Assoc 2005;68:204–9.
- Fernandez-Caldas E, Lockey RF. Blomia tropicalis, a mite whose time has come. Allergy. 2004;59:1161–4.
- Tsai JJ, Wu HH, Shen HD, Hsu EL, Wang SR. Sensitization to Blomia tropicalis among asthmatic patients in Taiwan. Int Arch Allergy Immunol. 1998;115:144–9.
- Lai CL, Shyur SD, Wu CY, Chang CL, Chu SH. Specific IgE to 5 different major house dust mites among asthmatic children. Acta Paediatr Taiwan 2002;43:265–70.
- Thind BB, Clarke PG. The occurrence of mites in cerealbased foods destined for human consumption and possible consequences of infestation. Exp Appl Acarol 2001;25:203–15.
- Musken H, Franz JT, Wahl R, Paap A, Cromwell O, Masuch G, Bergmann KC. Sensitization to different mite species in German farmers: clinical aspects. J Investig Allergol Clin Immunol 2000;10:346–51.
- Mullen GR, OConnor BM. Mites (Acari). In: Mullen GR, Durden LA. Medical and Veterinary Entomology. 2nd Ed. Academic Press (Elsevier Imprint), 2009.
- Jeong KY, Kim WK, Lee JS, Lee J, Lee IY, Kim KE, Park JW, Hong CS, Ree HI, Yong TS. Immunoglobulin E reactivity of recombinant allergen Tyr p 13 from Tyrophagus putrescentiae homologous to fatty acid binding protein. Clin Diagn Lab Immunol. 2005;12:581–5.
- Ferraroni NR, Oliveira S, Ferraroni JJ. Prevalence of Sensitization to the Storage Mite Acarus Siro in Middle-East of Brazil J Allergy Clin Immunol 2013;131:AB163.
- Pomés A, Chruszcz M, Gustchina A, Wlodawer A. Interfaces between allergen structure and diagnosis: Know your epitopes. Curr Allergy Asthma Rep. 2015 Apr;15(8):506.
Copyright @ 2017 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id