Basic Medical Research
Relative hypoxia and oxidative stress in spleen lymphocytes of immunized Balb/c mice as indicated by HIF-1α, HIF-2α, Nrf2 expression, and glutathione peroxidase activity
pISSN: 0853-1773 • eISSN: 2252-8083
https://doi.org/10.13181/mji.v27i4.2152 Med J Indones. 2018;27:223–8
Received: July 13, 2017
Accepted: August 9, 2018
Author affiliation:
1Master Program in Biomedical Science, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia
2Center of Hypoxia and Oxidative Stress Studies, Department of Biochemistry and Molecular Biology, Faculty of Medicine, Jakarta, Indonesia
Corresponding author:
Mohamad Sadikin
E-mail: sadikinmohamad@gmail.com
Background
Lymphocytes activated by immunization must increase their metabolism to meet the energy requirements for mitosis, differentiation, and protein synthesis, which may subject the cell to conditions of relative hypoxia and oxidative stress. This study was conducted to investigate the increase in the levels of transcription factors involved in both conditions.
Methods
Male Balb/c mice were divided into the following four groups, each consisting of six animals: the control and three experimental groups. The experimental groups were immunized by injection of 0.2 ml of 2% sheep red blood cells suspended in phosphate-buffered saline. Lymphocytes were harvested from the spleens of each group at time intervals of 24, 48, and 72 h postimmunization. The buffy coat from splenocytes was separated using Ficoll Histopaque as the medium. The lymphocytes were separated from adherent cells by incubating the purified splenocytes in microtubes for 2 h. Cells were lysed by three freeze–thaw cycles (−80°C and 37°C) and used to analyze the levels of HIF-1α and HIF-2α (mRNA and protein), Nrf2 (protein), and GPx activity.
Results
The treatment caused an increase in GPx activity and HIF-1α protein concentration 24 h post-immunization, whereas the HIF-1α mRNA levels remained static. Elevated Nrf2 protein levels were detected within 48 h after treatment. Meanwhile, the HIF-2α mRNA and protein levels increased within72 h after immunization.
Conclusion
Immunization with SRBC suspension induced relative hypoxia, elevated ROS, and oxidative stress in the lymphocytes as indicated by the increase in both HIF-1α and HIF-2α protein and mRNA levels, GPx activity, and Nrf2 protein levels.
Keywords
HIF-1α, HIF-2α, Nrf2, oxidative stress, relative hypoxia
Immunization is a method to elicit a specific defense response by exposing the organism to a foreign substance. The role of leukocytes is essential in this phenomenon. The immune response is categorized into nonspecific and specific immunity. Specific immunity, which is the desired outcome of every immunization, is mediated by mononuclear type of leukocytes, primarily macrophages or monocytes and lymphocytes, which are widely known as peripheral blood mononuclear cells (PBMCs). To elicit a specific immune response, both types of cells must perform various activities, i.e. migration, phagocytosis, synthesis of various cytokines, mitosis, and differentiation, and ultimately produce antibodies. Each activity requires vast amounts of energy, which is generally acquired through aerobic metabolism. If the oxygen supply does not match these metabolic demands, then the PBMCs involved in the immunization response are said to be in a state of relative hypoxia.i1–3
Almost all states of hypoxia disrupt several metabolic pathways in aerobic organisms. In response to hypoxic conditions, two transcription factors, hypoxia-inducible factor-1 (HIF-1) and hypoxia-inducible factor-2 (HIF-2), regulate a diverse number of genes that help aerobic organisms adapt to these harmful conditions.4,5 In addition, it has been reported that hypoxia leads to increased levels of reactive oxygen species (ROS).6 To counteract the ROS, cells possess a group of enzymes such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and glutathione reductase (GRed) that quench the free radicals. These enzymes are regulated by another transcription factor, the nuclear factor erythroid 2-related factor 2 (Nrf2).7–9
This study was conducted to explore whether the specific immune response mediated by lymphocytes in response to sheep red blood cell (SRBC) immunization leads to increased expression levels of HIF-1α, HIF-2α, and Nrf2 as well as GPx activity due to increased metabolic activity.
METHODS
This was an experimental study conducted at the Laboratory of Molecular Biology for Oxidative Stress, Department of Biochemistry and Molecular Biology, Faculty of Medicine, Universitas Indonesia, from October 2016 to April 2017. The animals were acclimatized for 2 weeks before treatment, followed by immunization and lymphocyte isolation. Splenocytes obtained from this procedure were used for analyzing the protein levels of HIF-1α, HIF-2α, and Nrf2 by enzymelinked immunosorbent assay (ELISA); mRNA expression analysis of HIF-1α and HIF-2α by quantitative real time reverse transcriptase PCR (qRT-PCR) method; and analysis of GPx activity by spectrophotometry.
Immunization and lymphocyte isolation
The animal model was obtained from Pusat Studi Satwa Primata, Bogor. This study was approved by the Ethics Committee of the Faculty of Medicine, Universitas Indonesia (No. 775/UN2. F1/ETIK/2016). Male Balb/c mice were randomly divided into the following four groups, each consisting of six mice: control, 24, 48, and 72 h. Mice from each group were injected intraperitoneally with 0.2 ml of 2% SRBC suspension. Furthermore, mice from each group were sacrificed after each period as indicated. Spleen samples were removed and homogenized with 500 μl phosphate-buffered saline (PBS). Splenocytes suspended in PBS were layered on top of Ficoll Histopaque 1.084 and centrifuged at 1,500 rpm for 30 min. The buffy coat was removed and washed with PBS. The isolated mononuclear cells were resuspended in PBS and incubated for 1–2 h to separate nonadherent cells from adherent cells. The nonadherent cell fraction that primarily contains lymphocytes was extracted and stored at −80°C until they were needed for analysis. Before analysis, the lymphocytes were lysed by three freeze–thaw cycles as follows: the frozen samples obtained from the −80°C chamber were directly placed in a 37°C incubator for 5 min for thawing, after which they were immediately placed in −80°C for 5 min, and this cycle was continued three times.10
Total protein quantification
To quantify total protein, 2 μl of the lysed samples was placed on a nanodrop plate, and the absorbance was measured at a wavelength of 280 nm. The absorbance was compared with the bovine serum albumine (BSA) standard curve to determine the total protein concentration.
Analysis of HIF-1α, HIF-2α, and Nrf2 proteins
The target proteins were quantified by ELISA11 antibodies obtained from Elabscience (mouse HIF-1α E-EL-M0687; mouse HIF-2α E-EL-M0786; mouse Nrf2 E-EL-M2607).
Analysis of HIF-1α and HIF-2α mRNA relative expression
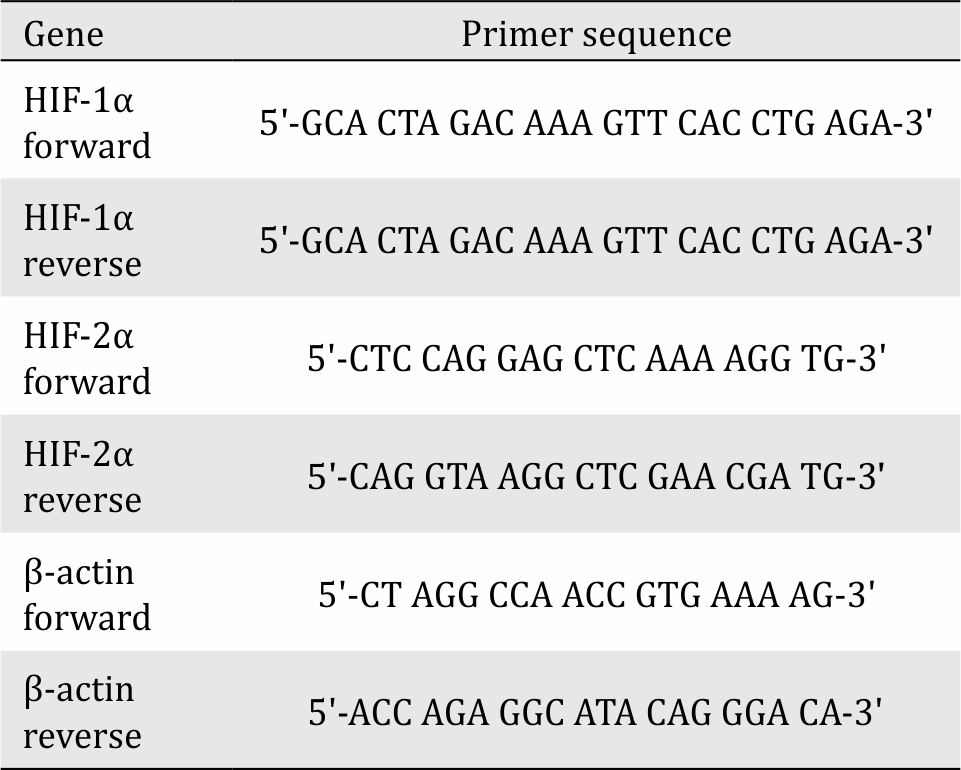
RNA from the cells was isolated by Tripure Isolation reagent (Ameritech, USA) using the β-actin housekeeping gene as control. The primers used in this process are listed in Table 1. The mRNA relative expression was analyzed using the procedure provided by One Step Kappa (KapaBiosystem). The PCR was run for 40 cycles. The annealing temperature was 52°C for the HIF-1α and HIF-2α primers and 54°C for the β-actin primer. Then, the Livak method12 was used to determine the amount of mRNA in the sample. All the primer sequences were obtained from NCBI (HIF-1α NM_001313919.1; HIF-2α NM_010137.3; β-actin NM_007393.5).
Table 1. HIF-1α, HIF-2α, and β-actin primer sequences
Analysis of GPx enzyme specific activity
The Ransel (Catalog no. RS505)13 kit assay was used to quantify the GPx activity. The principle of this quantification method involves the spectrophotometric determination of the nicotinamide adenine dinucleotide phosphate (NADPH) concentration remaining after the first and third minute of reaction, measured as absorbance at 340 nm. The amount of NADPH measured reflects the activity of GPx in converting reduced glutathione (GSH) into oxidized glutathione (GSSG).
Statistical analysis
The result obtained from ELISA was the absorbance of each sample, which was then converted into protein concentration through a standard curve. The amount of the desired protein was standardized to the total protein content. Data obtained from the GPx activity analysis were also in the form of absorbance, which were then converted into the amount of GPx enzyme using the equation pr ovided in the protocol13 and also standardized to the total protein content of each sample. Meanwhile, the fold change of target mRNA relative expression to the housekeeping gene was measured by the number of cycle threshold (Ct) and converted into relative expression by the Livak method.12 All data yielded from each assay were then evaluated for a significant difference by oneway analysis of variance (ANOVA) (for data that were normally distributed and homogenous), followed by a post hoc least significant difference (LSD) to determine the group that has a significant difference, and the Kruskal–Wallis test (for data that were not homogenous and normally distributed).
RESULTS
Expression of HIF-1α mRNA and protein
The relative mRNA expression of HIF-1α showed a significant increase in the 48 and 72 h groups (Kruskal–Wallis test, p=0.002). In contrast, the protein expression analyzed by one-way ANOVA showed a significant increase in the 24 h group (p=0.017) compared to the control group and a significant decrease in the 48 h (p=0.004) and 72 h (p=0.007) groups (Figure 1).
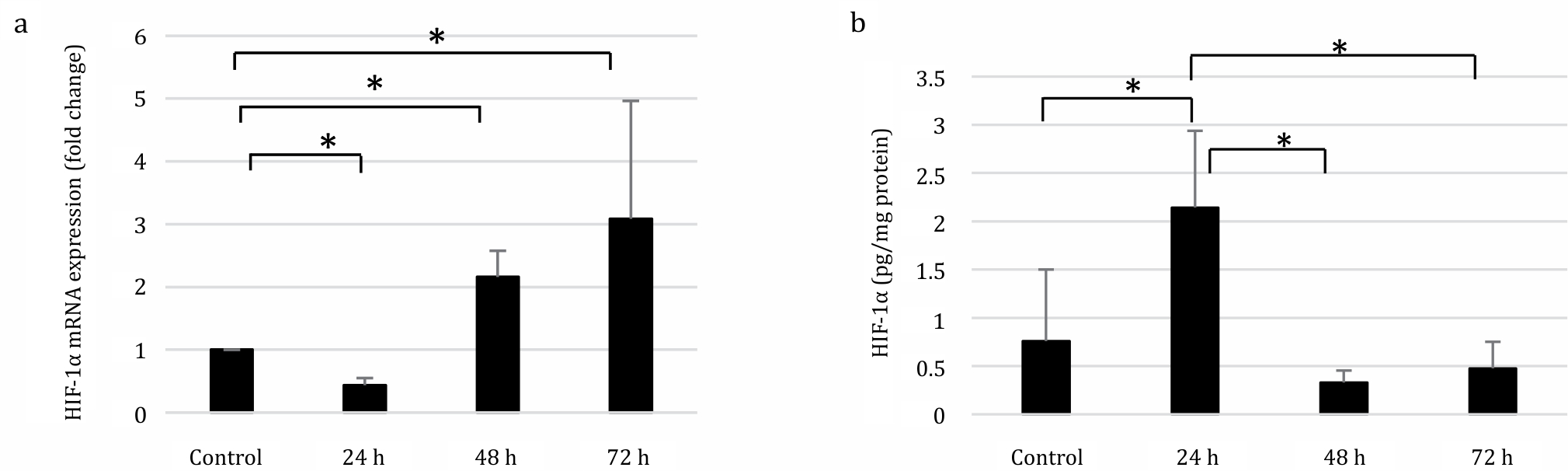
Figure 1. Expression of HIF-1α in SRBC-immunized mice after 24h. 48h, and 72h. (A) qRT-PCR was performed to deterimine the HIF-1α mRNA relative expression after SRBC exposure. mRNA relative expression was compared to that in control, whereas the control value was arbitrarily set to 1. HIF-1α mRNA relative expression was significantly increased two-fold after 48 and threefold 72 h of immunization relative to control group. (B) HIF-1α protein analysis was measured by ELISA. Immunization promotes hypoxia, indicated by the elevated levels of HIF-1α protein (2.143 pg/mg protein) after 24 h of SRBC exposure compare to control (0.759 pg/mg protein). *p<0.05
Expression of HIF-2α mRNA and protein
The relative mRNA expression of HIF- 2α was significantly increased at 24 and 72 h compared to the control group (Kruskal–Wallis test, p=0.034). The HIF-2α protein expression increased significantly in the 72 h group as analyzed by one-way ANOVA (p=0.036) (Figure 2).
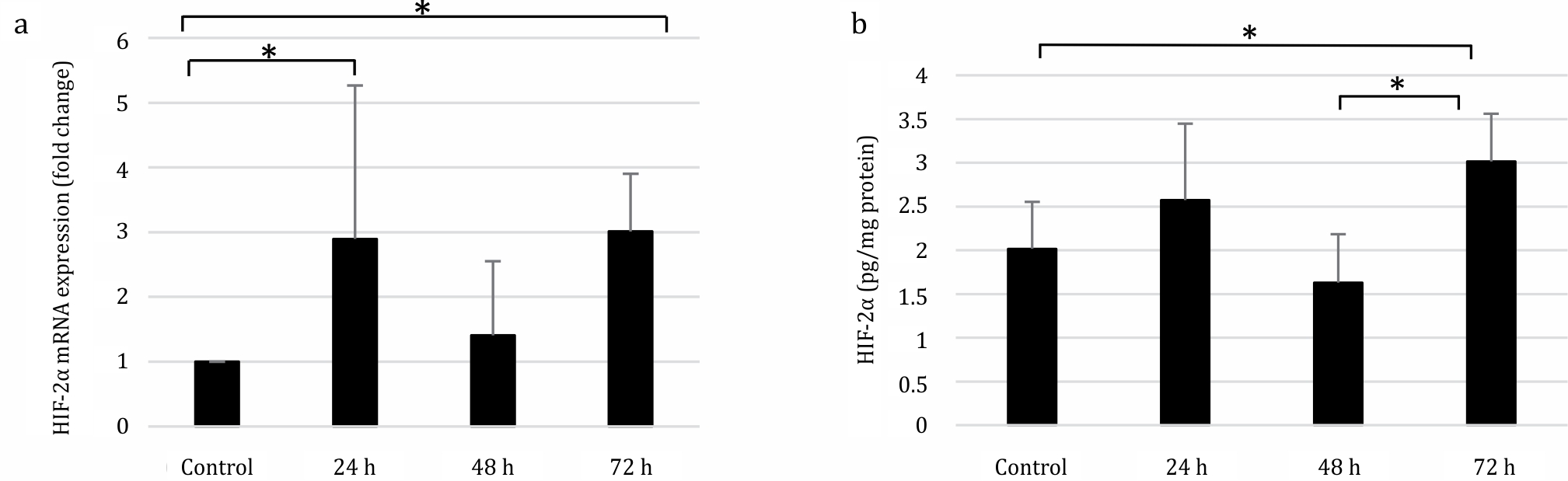
Figure 2. Expression of HIF-2α in SRBC-immunized mice. (A) mRNA relative expression was determined by qRT-PCR method. Relative mRNA expression was compared to that in the control group, which was set to 1. HIF-2α relative mRNA expression was increased three-fold both in the 24 and 72 h groups, compare control group. (B) HIF-1α protein expression was increased to 3.015 pg/mg protein after 72 h of treatment compare to control group (2.016 pg/mg protein) as evaluated by ELISA. *p<0.05
Nrf2 protein levels and GPx specific enzymatic activity
The Nrf2 protein levels were elevated in the 48 h group. Regarding the GPx specific enzymatic activity, there was a significant increase at 24 h post-immunization (one-way ANOVA, p=0.002) and a significant decrease after 48 h (p=0.000) and 72 h (p=0.001) of immunization (Figure 3).
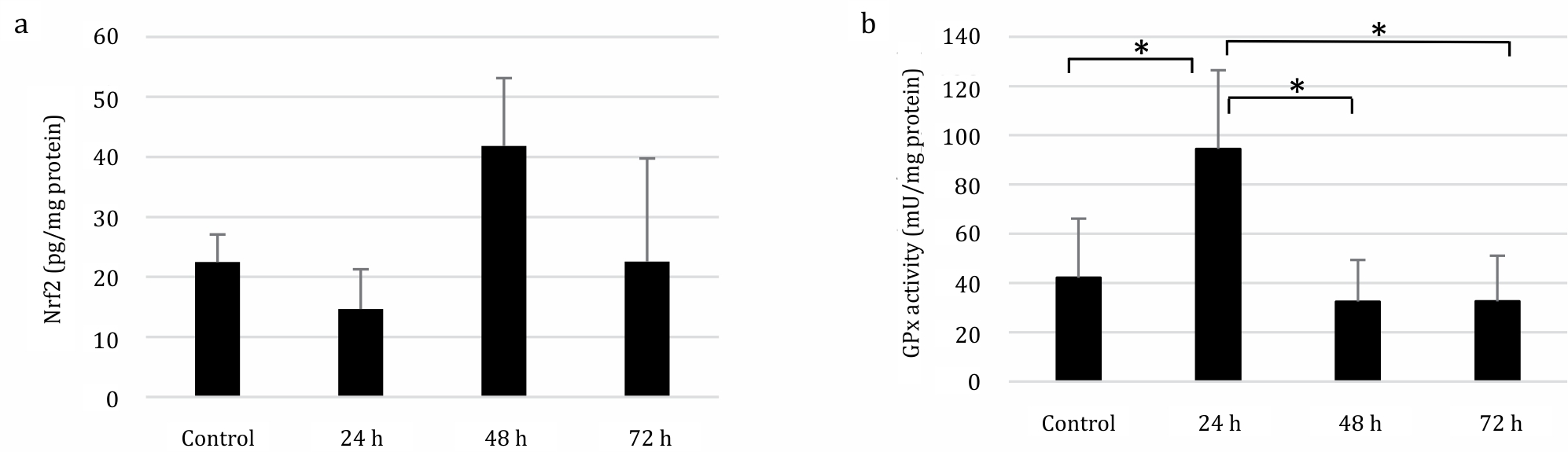
Figure 3. Nrf2 protein expression and GPx activity in SRBC-immunized mice. (A) Nrf2 protein expression was measured by ELISA, which showed an increase (41.8 pg/mg protein) 48 h post-immunization compare to control group (22.49 pg/mg protein). (B) Spectrophotometry was used to determine the GPx activity, which showed an increase (94.5 mU/mg protein) after 24 h of immunization compare to control group (42.14 mU/mg protein). *p<0.05
The results of this study indicated that SRBC immunization induced a state of relative hypoxia in the lymphocytes after 24 h, as indicated by the increase in HIF-1α protein levels. This phenomenon is caused due to the stabilization of the protein rather than de novo synthesis as indicated by the low mRNA levels. On the other hand, the relative expression of mRNA was elevated after 48 and 72 h of antigen exposure, which may be the result of de novo compensation of HIF-1α levels in response to hypoxic conditions.3,14
Based on these results, not all mRNA was translated to its corresponding protein or that it was translated but quickly degraded. Alternatively, report shown that the HIF-1α protein expression can be regulated by its own downstream target gene.15 Thus, the elevation of HIF- 1α protein may due to the transcription of its own target gene.
The analysis of HIF-2α mRNA and protein levels showed that the relative expression of mRNA was elevated 24 and 72 h post-immunization. Meanwhile, the protein levels increased 72 h after immunization. The increase in mRNA levels in the 24 h group is most probably a response to relative hypoxia. However, the mRNA was not translated immediately into protein, which may be due to the high levels of HIF-1α protein.5,14 The HIF-2α protein levels were increased after 72 h of immunization, which could be attributed to the role of HIF-2α protein in prolonged hypoxia; thus, the 72 h period after immunization can be categorized as chronic hypoxia.14,16
Previous studies have demonstrated that DNA synthesis in PBMCs reaches its maximum level at 48 h after stimulation with phytohemagglutinin A (PHA).17,18 In our study, we applied an immunization procedure using a complex antigen, i.e. SRBC suspension. A complete immune response includes not only the mitosis of both T and B lymphocytes but also the synthesis of immunoglobulins by B lymphocytes after their differentiation into plasmocytes. It is well known that plasmocytes can be easily observed under a microscope 72 h after immunization. At this stage, plasmocytes synthesize and produce antibodies, which require a vast amount of energy. At 72 h after immunization, there is a peak in HIF-2α levels, which indicates a state of chronic hypoxia. In addition, HIF-2α is primarily expressed by differentiated cells.2,19
HIF-2α protein is known for its role in supporting the progression of the cell cycle, while HIF-1α protein is known for its role in supporting the cell to enter the G0 phase (arrest). Elevated concentrations of HIF-1α protein 24 h after immunization may be a response to oxygen scarcity. After 48 and 72 h, HIF-2α protein initiates angiogenesis, vasodilation, and erythropoiesis to meet the lymphocytes’ energy requirement for differentiation.19
Another study using SRBC-immunized mice was conducted on macrophages, which demonstrated that macrophages had increased levels of ROS as a result of the immune response to SRBCs.20,21 Moreover, in our previous study, we demonstrated that rats that were placed under hypoxic condition for an extended period of time experienced oxidative stress as indicated by the increase in malondialdehyde (MDA) levels and a decrease in GSH levels.22 We concluded that this paradoxical condition may be due to mitochondrial leakage and the high level of xanthine dehydrogenase activity. In the present study, we hypothesized that healthy lymphocytes under normoxic conditions, when immunized, will concurrently exhibit hypoxia and oxidative stress. We measured the levels of the transcription factor Nrf2, which regulates various antioxidant proteins such as GPx.
This study also confirmed that there was a peak in free radical production in the 24 h group, which could be due to increased metabolism of activated lymphocytes, exhibited by the elevation of GPx specific enzymatic activity. The Nrf2 protein levels were not increased during the 24 h time interval, presumably because the increase in free radical production can still be counteracted by existing GPx. However, 48 h after immunization, the lymphocytes were predicted to become active and required more energy, thereby leading to oxidative stress and ultimately the increased expression of Nrf2 protein.
The Nrf2 protein regulates the transcription of not only GPx but also other antioxidant enzymes such as catalase (CAT) that has the same substrate as GPx.7,8,23 In the 48 h group, the elevated concentrations of Nrf2 might also lead to the transcription of CAT. Hence, the high concentration of free radicals was neutralized by CAT and the GPx enzyme specific activity also decreased.8
Lymphocytes also experienced oxidative stress as indicated by an elevation of Nrf2 protein levels after 48 h of immunization. This condition is a result of increased levels of oxidative respiration required to meet the continuous energy demands under hypoxic conditions. Therefore, the findings of this study suggest that hypoxia occurs before oxidative stress.
In conclusion, immunization of Balb/c mice with SRBC induced relative hypoxia in the lymphocytes 72 h after immunization, as indicated by the increasing levels of HIF-1α and HIF-2α mRNA and protein. Actively proliferating and differentiating lymphocytes produce high amounts of ROS after 24 h of immunization. These ROS were neutralized by the GPx enzyme.
Conflicts of Interest
Sri W.A. Jusman is one of the editorial board members, but was not involved in the review or decision process of the article.
Acknowledgment
This research was funded partly by Hibah PUPT UI 2015 Nomor 1684/UN2.RI2/ HKP.05.00/2015.
REFERENCES
- Gilkes DM, Semenza GL. Role of hypoxia-inducible factors in breast cancer metastasis. Futur Oncol. 2013;9(11):1623–36.
- Masoud GN, Li W. HIF-1 α pathway: role, regulation and intervention for cancer therapy. Acta Pharm Sin B. 2015;5(5):378–89.
- Liu Z, Semenza GL, Zhang H. Hypoxia-inducible factor 1 and breast cancer metastasis. J Zhejiang Univ Sci B. 2015;16(1):32–43.
- Gilkes DM, Bajpai S, Chaturvedi P, Wirtz D, Semenza GL. Hypoxia-inducible Factor 1 (HIF-1) promotes extracellular matrix remodeling under hypoxic conditions by InducingP4HA1, P4HA2,andPLOD2 expression in fibroblasts. J Biol Chem. 2013;288(15):10819–29.
- Stavik B, Espada S, Cui XY, Iversen N, Holm S, Mowinkel M, et al. EPAS1/HIF-2 alpha-mediated downregulation of tissue factor pathway inhibitor leads to a pro-thrombotic potential in endothelial cells. Biochim Biophys Acta. 2016;1862(4):670–8.
- Jusman SW, Halim A. Oxidative stress in liver tissue of rat induced by chronic systemic hypoxia. Makara Kesehatan. 2009;13(1):34–8.
- Kim SG, Lee WH, Kim YW. Nrf2. Encyclopedia of signaling molecules. In: Choi S, editor. Springer Science+Business Media; 2012. p. 80–5.
- Fuse Y, Kobayashi M. Conservation of the Keap1-Nrf2 system: an evolutionary journey through stressful space and time. Molecules. 2017;22(3).
- Jaramillo MC, Zhang DD. The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Gene Dev. 2013;27(20):2179–91.
- Walker JM. The Protein Protocols Handbook. 3rd ed. New York: Springer-Verlag New York, LCC; 2009.
- Rodwell VW, Bender DA, Botham KM, Kennelly PJ, Weil PA. Harper’s Illustrated Biochemistry. 30th ed. New York: McGraw Hill; 2015.
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C(T)) method. Methods. 2001;408:402–8.
- Ransel Glutathione Peroxidase Manual [Internet]. UK: Randox Laboratories, Ltd. 2005 [cited Oct 2017 ]. p. 1–2. Available from: https://www.sanyo-si.com/wp-content/ uploads/e28aa06ef42fe6393a72a6f9052e365d. pdf
- Florczyk U, Czauderna S, Stachurska A, Tertil M, Nowak W, Kozakowska M, et al. Opposite effects of HIF-1 α and HIF-2 α on the regulation of IL-8 expression in endothelial cells. Free Radic Biol Med. 2011;51(10):1882–92.
- Deudero JJ, Caramelo C, Castellanos MC, Neria F, Fernández- Sánchez R, Calabia O, et al. Induction of hypoxia-inducible factor 1α gene expression by vascular endothelial growth factor*. J Biol Chem. 2008;283(17):11435–44.
- Ratcliffe PJ. HIF-1 and HIF-2 : working alone or together in hypoxia ? J Clin Invest. 2007;117(4):399–402.
- Mustofa S. Penghambatan aktivitas anhidrase karbonat menurunkan proliferasi sel mononukleus darah tepi (SMDT) Manusia yang distimulasi dengan phytohaemagglutinin dan interleukin-2. Universitas Indonesia; 2016. Indonesian.
- Firakania C. Penghambatan proliferasi sel mononukleus darah tepi (SMDT) manusia oleh avidin, pascastimulasi fitohemaglutinin (PHA) dan Interleukin-2 (IL-2). Universitas Indonesia; 2015. Indonesian.
- Gordan JD, Bertovrt JA, Hu C, Diehl JA, Celeste MS. HIF-2 α promotes hypoxic cell proliferation by enhancing c-Myc transcriptional activity. Cancer Cell. 2007;11(4):335–47.
- Asikin H. Respon makrofag limpa mencit pasca imunisasi: kajian terhadap ekspresi hypoxia-inducible factor-1a, hif-2a, sitoglobin dan peroxisome proliferator activated receptor gamma coactivator-1a. Universitas Indonesia; 2016. Indonesian.
- Sarsanti PAN. Hipoksia pada makrofag peritoneum mencit Balb/c yang diimunisasi: kajian terhadap HIF-1 α , HIF-2 α , Sitoglobin, dan PGC-1 α. Universitas Indonesia; 2017. Indonesian.
- Sadikin AH. Stres oksidatif pada hati tikus yang diinduksi hipoksia sistemik. Universitas Indonesia; 2008. Indonesian.
- Buendia I, Michalska P, Navarro E, Gameiro I, Egea J, León R. Nrf2-ARE pathway: an emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol Therapeut. 2016;157:84–104.
Copyright @ 2018 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id
