
Section Abstract Introduction Case Illustration Discussion Conflict of Interest Acknowledgment References
Case Report
Reducing severe spasticity in cerebral palsy following meningoencephalitis by botulinum toxin
pISSN: 0853-1773 • eISSN: 2252-8083
http://dx.doi.org/10.13181/mji.v26i1.1297 Med J Indones. 2017;26:76–80
Received: November 17, 2016
Accepted: Februari 17, 2017
Author affiliation:
Department of Pediatric Neurology, Airlangga University, Surabaya, Indonesia
Corresponding author:
Prastiya I. Gunawan
E-mail: prastiya_ig@yahoo.co.id
A 3-year-old boy diagnosed with diplegic cerebral palsy had received Botulinum toxin injection to reduce severe spasticity. There was an improvement of muscles tone and motor function including better ability of limb flexion of the knees and hip, adduction of the hip, flexion of the toes, and plantar flexion of the foot. No side effect was observed after the procedure.
Keywords
botox, cerebral palsy, spasticity
Cerebral palsy (CP) is the most common physical disability disease in childhood. The motor impairment symptoms are obligatory for the diagnosis, but a broad range of neurological deficits can be found as well.1,2 The brain injury that results hypoxic ischemic in developing brain can occur anytime from gestation to early childhood, which might be responsible for CP pathophysiology.2
A wide variety of medication has been used to treat CP. Diazepam and baclofen are commonly used for spasticity as they have narrow therapeutic safety. Yet, careful monitoring is required to prevent over dosage. Botulinum toxin (Botox) has been recommended as a therapeutic option for childhood spasticity in CP (recommendation A). It provides focal, controlled muscle weakness and reduction of spasticity. The best evidence of Botox usage is for equinus varus treatment.3,4 The effects of Botox can be seen on spasticity, range of motion (ROM), gross motor function, and gait improvement.5,6
The aim of this paper is to report the effectivity of botulinum toxin in the management of severe spasticity in a child with spastic diplegia CP following meningoencephalitis.
CASE ILLUSTRATION
A 3-year-old boy, who was a cerebral palsy child, was referred to a Pediatric Emergency Department at the Dr. Soetomo Hospital, Surabaya, Indonesia on June, 12 2014 because of continue spasms. He had a history of unconsciousness and two-month-period of fever before the referral. Both upper and lower extremity were spastic. An episode of convulsion was recorded in which both arms extended and hands gripped, with his gaze upward, lasted for about three minutes.
The neurological examination revealed spastic diplegia of four extremities with no lateralization. All extremities were atrophy. The legs were contracture, with scissor-like appearance and feet were both plantar-flexed. The neck and back muscles were rigid eventually caused scoliosis posture (Figure 1).
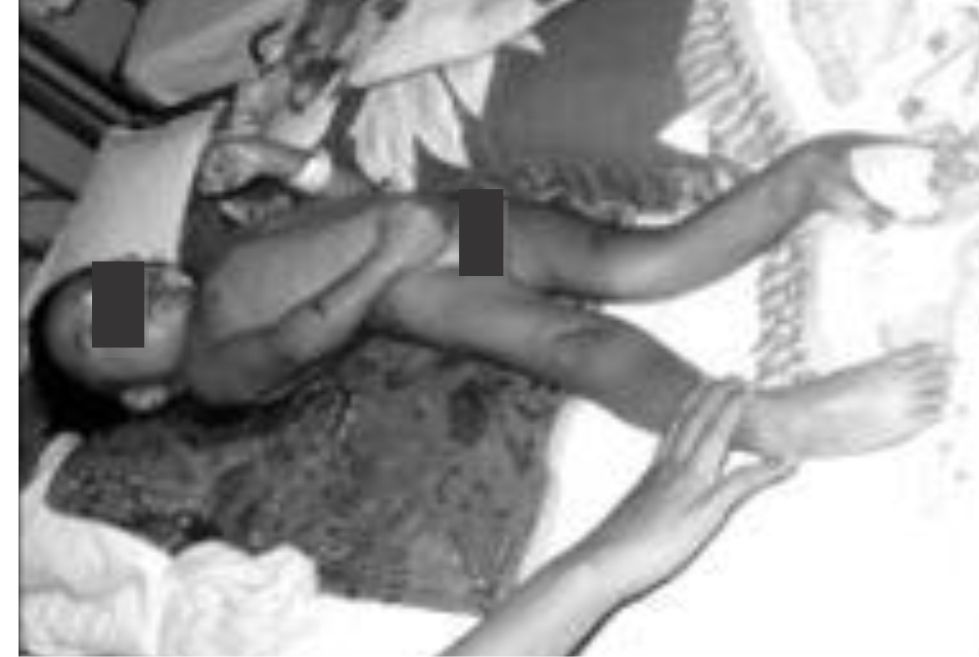
Figure 1. Posture of a child with spastic diplegia cerebral palsy
The electroencephalogram (EEG) recording described generalized diffuse encephalopathy and multiregional epileptiform activities. Head computed tomography (CT) scanning without contrast revealed meningoencephalitis, parietal and frontal subdural hygroma, and bilateral maxillary sinusitis. He was diagnosed as meningoencephalitis and cefepime, baclofenâ, and valproic acid were instituted. The patient’s condition improved within few days. The temperature returned to normal, and culture taken from three location was sterile. During three weeks of hospitalization, the spasticity reduced. The child was able to open his grasp although the lower extremity did not significantly improved. There was no more episode of stiffness with upward gaze. The patient was consulted to rehabilitation department to maintain the muscles strength.
On week four, botulinum toxin was injected for his lower extremities to reduce spasticity. A dose of 100 units (U) was given, divided into 10 U for each rectus femoris, vastus intermedia, tibialis posterior, soleus muscles, and gastrocnemius muscles. After the procedure, both of the lower extremities were bandaged from thigh to ankle for a week before it was released.
Four weeks after the procedure, the boy was able to sit with his mother support. Both ankles were still bandaged to maintain those dorsoflexion. The scoliosis posture and scissor legs were also improved. On the fifth week after injected with Botox, the bandage was released, and special shoes were given to maintain the ankles on flexion position. The Ashworth scale to measure spasticity improved from grade 4 to 2.
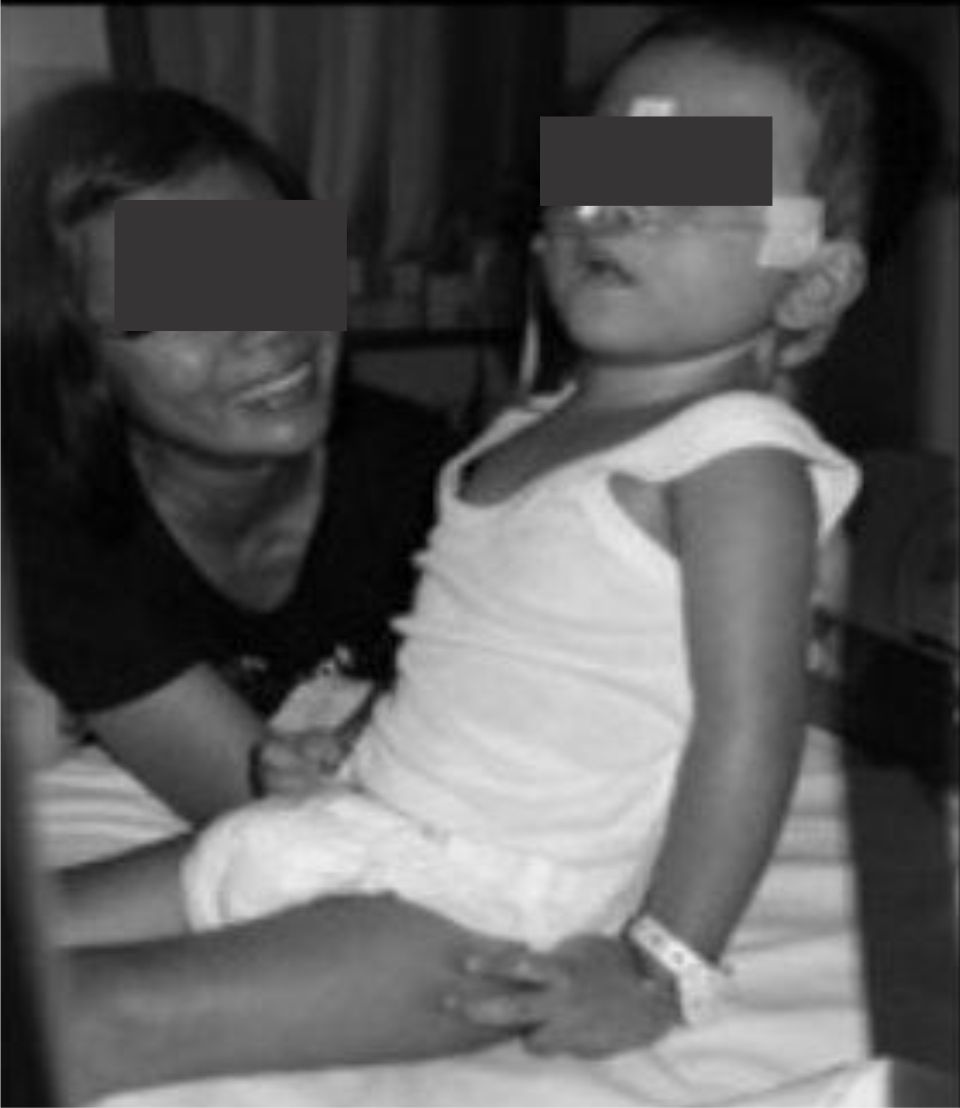
Figure 2. Four weeks after Botox injection: the child was able to sit with better flexion of his knee joints
Physical therapy continued to exercise the muscles. Soleus muscles and lower arm muscles which atrophied initially and improved after two months of therapy. The physiological reflexes of lower extremity were also occured. The parents had already informed a consent for publishing the case.

Figure 3. Five weeks after Botox injection: flexion of the ankles almost 90° with special shoes
DISCUSSION
Physical therapy was instituted to preserve muscles strength from the beginning. To justify an intervention, there must be a purpose for treating spasticity that is meaningful to patient and their caregivers. Due to the major deformity on lower limb (scissor leg and spastic equinus), we opted for the Botox injection. Botulinum toxin is regularly used for a goal-directed focal management of spasticity and dystonia for children with cerebral palsy. In our case, the patient was non-ambulatory CP that suffered from severe spasticity following intracranial infection and also epilepsy.7 One evidence-based review recommended Botox injection of the calf muscles as a treatment option for equinus varus deformity in children with CP.3 For localized spasticity that warrants management, botulinum toxin type A is suggested as an effective and commonly safe treatment (level A based on American Academy of Neurology guidelines).6 This Botox injection is a part of management algorithm for diplegia.8–11 In this case, Botox injection was instituted to rectus femoris, vastus intermedius, tibialis posterior, gastrocnemius, and soleus of both lower extremity. The satisfaction rate of Botox injection was approximately 69.2%.12 The goal of this treatment may include simple aims such as reducing pain, increasing range of motion, and preventing of secondary medical complications like contractures.12
A dose of 100 U was given to the patient, divided into 10 U for each location. Choosing an appropriate injection dose for children is often based on patient’s body weight. Many studies use different dosages to treat spasticity in children with diplegia, hemiplegia, and tetraplegia CP. The dosage can be given as 15 U each muscles or given in certain unit per body weight (12 U/kg or 5–28 U/kg).9 Another way to give Botox is to give 100 U or 200 U per treated leg. Bjornson applied 12 units/kg of Botox-A in 33 children with spastic diplegia in gross motor function classification system (GMFCS) I-III, and improvement was found in performance goals at 12 weeks and in maximum voluntary torque and gross motor function at 24 weeks.6
Improvements of muscles tone and motor function in this child include a better flexion of the hip and knees, an adduction of the hip, a flexion of the toes, and a plantar flexion of the feet. These muscles tone’s improvements could be evaluated using the Ashworth Scale. This is a simple method that has been widely used as a clinical quantifiable of spasticity and has a good face validity. The patient showed significant improvement of the Ashworth Scale from 4 (affected part rigid in flexion or extension) to 2 (marked increase in muscle tone, manisfested by catch in the middle range and resistance throughout the remainder of the range of motion, but affected part is easily removed). The correction of scoliosis posture, knee joints, and scissor legs were due to its mechanism of action to block acetylcholine release at neuromuscular junctions.3 A similar case of gastrocnemius spasticity in 4-year-old child who was also given Botox injection showed improvements of hip and knee joints range of motion (ROM), ankle kinematics and kinetic, and a better ankle position with amplification of its capacity of propulsion during terminal stance.10
The adverse effects associated with administration of Botox could be a diffusion of the toxin from the attempted sites of action that can lead to unwanted inhibition of transmission at neighboring nerve endings. Fortunately, these did not obvious in this child. The patient did not experience dysphagia, hypotonia, poor feeding, ptosis, and generalized weakness as reported in literature. Risk of developing systemic effects does not seem to be related to dose based on body weight. It may be more likely related to total injection dose and frequency.13 Some patients required ibuprofen for muscle soreness at injection site and few of them decreased their activity level for 24 hour-postinjection. 13
Other treatments for severe spasticity that currently ongoing are the functional posterior rhizotomy (FPR) and the intratechal baclofen (IB). FPR surgery is carried out in the cauda equine, and neurophysiological procedures are used to judge which nerve root should be cut. The outcome of FPR is favorable in the long term follow up. In IB, since it is a surgery performed to implant foreign bodies, a special attention should be paid to avoid perioperative complication such as meningitis and mechanical failure.11 When FPR with intensive physical therapy was compared to intensive physical therapy alone, the former showed better functional improvement and was more effective to reduce spasticity. Both of recommended treatments were not performed due to availability of the device, precious value, side effects such as infection and transformation of spinal curvature. Botox injection is less invasive compared with the FPR and IB infusion treatment.11
In conclusion, severe spasticity is a major challenge for rehabilitation of children with CP. Botox injection improves muscle performance in a child with CP severe spastic diplegia following meningoencephalitis. It improved a scissoring and reduced leg spasticity. Further evaluation is necessary to determine ambulatory status. The combination of these treatments is the key to successful treatment of childhood severe spasticity.
Conflicts of Interest
The authors affirm no conflict of interest in this study.
Acknowledgment
None.
REFERENCES
- Kakooza-Mwesige A, Forssberg H, Elliasson A, Turnwine JK. Cerebral palsy in children in Kampala, Uganda: clinical subtypes, motor function and co-morbidities. BMC Res Notes. 2015;8:166.
- Novak I, Mclntyre S, Morgan C, Campbell L, Dark L, Morton N, et al. A systematic review of interventions for children with cerebral palsy: state of the evidence. Dev Med Child Neurol. 2013;55(10):885–910.
- Whelan MA, Delgado MR. Practice parameter: pharmacologic treatment of spasticity in children and adolescents with cerebral palsy (an evidence-based review): report of the quality standards subcommittee of the american academy of neurology and the practice committee of the child neurology society. Neurology. 2010;75(7):669.
- Delgado MR, Hirtz D, Aisen M, Ashwal S, Fehlings DL, McLaughlin J, et al. Practice parameter: pharmacologic treatment of spasticity in children and adolescents with cerebral palsy (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology. 2010;74(4):336–43.
- Pavone V, Testa G, Restivo DA, Cannavò L, Condorelli G, Portinaro NM, et al. Botulinum toxin treatment for limb spasticity in childhood cerebral palsy. Front Pharmacol. 2016;7(29):1–6.
- Bjornson K, Hays R, Graubert C, Price R, Won F, McLaughlin JF, et al. Botulinum toxin for spasticity in children with cerebral palsy: a comprehensive evaluation. Pediatrics. 2007;120(1):49–58.
- Li SY, Qian XG, Zhao YL, Fu WJ, Tan XR, Liu ZH. Risk factors for interictal epileptiform discharges on electroencephalogram in children with spastic hemiplegic cerebral palsy. Zhongguo Dang Dai Er Ke Za Zhi. 2015;17(12):1338–41. Chinese.
- Zhu Dn, Wang MM, Wang J, Zhang W, Li HZ, Yang P, et al. Effect of botulinum toxin A injection in the treatment of gastrocnemius spasticity in children aged 9-36 months with cerebral palsy: a prospective study. Zhongguo Dang Dai Er Ke Za Zhi. 2016;18(12):123–9. Chinese.
- Choi JY, Jung S, Rha D, Park ES. Botulinum toxin type A injection for spastic equinovarus foot in children with spastic cerebral palsy: effects on gait and foot pressure distribution. Yonsei Med J. 2016;57(2):496–504.
- Cimolin V, Galli M, Crivellini M, Albertini G. Quantitative effects on proximal joints of botulinum toxin treatment for gastrocnemius spasticity: a 4-year-old case study. Case Report Med. 2009;10(985717):1–4.
- Morota N, Ihara S, Ogiwara H. Neurosurgical management of childhood spasticity: functional posterior rhizotomy and intratechal baclofen infusion therapy. Neurol Med Chir. 2015;55(8):624–39.
- Molenaers G, Van Campenhout A, Fagard K, De Cat J, Desloovere K. The use of botulinum toxin A in children with cerebral palsy, with a focus on the lower limb. J Child Orthop. 2010;4(3):183–95.
- Crowner BE, Torres-Russotto D, Carter AR, Racette BA. Systemic weakness after therapeutic injection of botulinum toxin A: a case series and review of the literature. Clin Neuropharmacol. 2010;33(5):243–7.
Copyright @ 2017 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id