
Section Abstract Introduction Methods Results Discussion Conflict of Interest Acknowledgment References
Basic Medical Research
Antiviral effect of Archidendron pauciflorum leaves extract to hepatitis C virus: An in vitro study in JFH-1 strain
pISSN: 0853-1773 • eISSN: 2252-8083
https://doi.org/10.13181/mji.v27i1.2189 Med J Indones. 2018;27:12–8
Received: August 3, 2017
Accepted: January 12, 2018
Author affiliation:
1 Research Centre for Chemistry, Indonesian Institute of Science (LIPI), Kawasan PUSPIPTEK, Tangerang, Indonesia
2 Department of International Health, Kobe University, Graduate School of Health Sciences, Kobe, Japan
3 Department of Microbiology, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia
4 Department of Oral Vaccine and Drug Development, Kobe University, Graduate School of Health Sciences, Kobe, Japan
Corresponding author:
Muhammad Hanafi
E-mail: hanafi124@yahoo.com
Background
Hepatitis C virus (HCV) is a leading cause of chronic liver diseases. Drug resistance to the regimen is also increasing. Hence, there is a need for new anti-HCV agents that are less toxic and more efficacious. The aim of this study is to evaluate the possibility of A. pauciflorum extracts can be a antiviral drug.
Methods
Huh-7it cells were infected with the HCV genotype 2a strain JFH-I in the presence of methanol extracts of Archidenron pauciflorum. The methanol extract further partition used n-hexane, ethyl acetate, n-butanol, and water showed in which butanol extracts exerted the strongest IC50 (6.3 g/ml). Further, the butanol fraction was fractionated and yielded into 13 fractions.
Results
The methanol extract of the leaves of A. pauciflorum exhibited concentration dependent inhibition against the JFH1 strain of HCV genotype 2a with an IC50 is 72.5 μg/ml. The butanol fraction exhibited the highest anti-HCV activity with an IC50 is 6.3 μg/ml. The butanol fraction was fractionated which yielded 13 fractions. Fractions 5 and 13 exhibited high anti-HCV activities with IC50 is 5.0 μg/ml and 8.5 μg/ml and a time-of-addition study demonstrated that fraction 5 inhibited viral infection at the post-entry step, whereas fraction 13 primarily inhibited the viral entry step.
Conclusion
The extract A. pauciflorum can be used as a herbal-based antiviral drug.
Keywords
Archidendron pauciflorum, HCV, JFH-1
Hepatitis C infection disease caused by the hepatitis C virus (HCV) primarily affects the function of liver. HCV is a major cause of chronic liver diseases and linked to the development of cirrhosis and hepatocellular carcinoma. Approximately 130–200 million people worldwide are infected with HCV,1 which is spread primarily by blood-to-blood contact which associated with intravenous drug use, contaminated medical equipment, transfusion, and organ transplants. Boceprevir or telaprevir plus pegylated interferon alpha (IFN-α) and ribavirin are the standard therapies for patients with chronic HCV infections. This triple combination therapy increases the sustained viral, with the response rates up to 75% in patients with HCV genotype 1 infections, which respond relatively poor to IFN-α therapy.2 However, it is still important to search for new and effective anti-HCV drugs to increase sustained viral responses as well as reduce the treatment side effects and costs.
Medicinal plants are attractive and important resources of bioactive compounds.3 Natural molecules derived from medicinal plants and natural products have been identified as anti-HCV agents in recent years.4–12 Among them, silymarin, ladanein, and (-)-epigallocatechin have been shown to be bioactive via oral administration in mice, healthy individuals, or HCV-infected patients,13–15 representing potential therapeutic use in clinical trials.
Djengkol is the common name of Archidendron pauciflorum. It is also called Abrema jiringa Kosterm, Albizia jiringa (Jack) Kurz, Albizia lucida sensu auct. Archidendron pauciflorum (Benth.) I. C. Nielsen, Feuileea jiringa Kuntz, Pithecellobium jiringa (Jack) Parin etc.16 A. jiringa is a leguminous tree plant belonging to the family of Fabaceae. This plant has been used as a traditional medicine for a range of ailments and consumed as a raw vegetable in Malaysia.17 Some chemical constituents is djenkolic acid (sulfurcontaining amino acid), from leaves, have been reported containing five flavan-3-ol derivatives and new flavan-3-ol galates, gallocatechin-3’ and 4’-O-gallates, gallocatechin7,3’ and 7,4’di-O-gallates. Procyanidinds-B3 and B-4, prodelphinidine B-1. Methanol extract leaves of A. jiringa active as antibacterial were mostly active for Staphylococcus aureus, S. epidermidis, and Microsporum gypsum.18 The shoot of A. jiringa has been found to have high polyphenolic contents (>150 g gallic acid equivalents/mg dried plant) and antioxidant activities measured using ferric reducing antioxidant power (FRAP) method, also have high potent to 2,2-diphenyl- 1-picrylhydrazyl(DPPH) scavenging activity.19,20 The beans of this plant are used as food sources in Indonesia and contain djenkolic acid and sulfur. Those elements can inhibit the pests as well as vitamin C and poly phenolic compounds, which have potential anti-oxidant activities.21 Plants of the Fabaceae family have been reported to exhibit various anti-viral activities. For example, glycyrrhizic acid from Glycyrrhiza uralensis Fisch was reported to exert anti-viral activities against coxsackievirus A16 and enterovirus 71, which are the most common causative agents of hand, foot, and mouth disease.20 Extracts from Acacia confusa, were shown to exhibit anti-HCV activities.21–24 A sulfate prepared from galactomannan derived from Mimosa scabrella was reported to exert in vitro anti-herpetic and anti-rotaviral activities.25 However, the possible anti-viral activities of A. pauciflorum extracts have not yet been reported.
In the course of our screening program of the anti-HCV activities of Indonesian medicinal plants, we observed the potential of the methanol crude extract and butanol fraction from the leaves of A. pauciflorum as anti-viral activities against HCV. Bioactivity-guided fractionation was performed and the mode of action of these fractions with anti-HCV activities was analyzed.
METHODS
Study Design
This was an in vitro study in human hepatocyte culture, Huh-7it cells using hepatitis C virus genotype-2a JFH-1 strain. Huh-7it cells were used to propagate HCV. All of the experiments were conducted in triplicate.
Collection of A. pauciflorum, extraction of crude extracts, and preparation of fractions
A. pauciflorum leaves used in this study were obtained from the Research Center for Chemistry, Indonesian Institute of Sciences (LIPI), Kawasan Puspiptek Serpong, Indonesia. The leaves samples were verified by botanical researchers at Herbarium, Research Center for Biology, LIPI, Cibinong, Indonesia. A herbarium specimen was deposited in the Research Center for Chemistry-LIPI. A. pauciflorum leaves were dried, pulverized, and subsequently macerated in 100% methanol for 24 h. The extracts were filtered and the residue was resuspended in fresh 100% methanol. This maceration process was repeated over three days. The combined filtrates were evaporated at room temperature, not exceeding 40°C. The extract (80 g) was initially partitioned with n-hexane, ethyl acetate, n-butanol, and water to obtain a hexane fraction (18.2 g), ethyl acetate fraction (3.0 g), butanol fraction (30.0 g), and water fraction (25.3 g). Based on bioactivity results, the butanol fraction (2.5 g) was subjected to silica gel 60 column chromatography (0.063– 0.200 mm; Merck, Darmstadt, Germany) with n-hexane: ethyl acetate and ethyl acetate: methanol by gradient elution, which yielded 13 subfractions (fraction number 1–13).
Sample preparation
The methanol crude extracts and subfractions were dissolved in DMSO to obtain stock solutions at concentrations of 100 and 50 mg/ml, respectively. The stock solutions were stored at −30°C until used.
Cell culture and virus preparation
Huh7it-1 is a well differentiated hepatocyte derived from cellular carcinoma cell line that was originally taken from a liver tumor in a 57 year-old Japanese male in 1982. The cell line was established by Nakabayashi and Sato J.25
The human adapted variants of the HCV strain JFH-1 (JFH1a)26 were inoculated into the Huh7it-1 cells. Culture supernatants were collected at 3–5 days post infection (p.i.), concentrated using Amicon 100K centrifugal filters, and stored at −80°C. Huh7it-1 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (Biowest, Nuaille, France), 100 μg/ml kanamycin (Sigma-Aldrich, St. Louis, MO, USA), and non-essential amino acids (Invitrogen). Cells were grown at 37°C in an atmosphere of 5% CO2.
Virus titration
Cells (2.4×104 cells/well) were placed in a 96-well plate 1 day before the virus infection. Culture supernatants were serially diluted 10-fold with medium in a 96-well plate, and 50 μL of viruses were adsorbed to the cells for 4 h at 37°C. Then, the cells were incubated with a medium containing 0.4% methylcellulose (Sigma-Aldrich) for 40 h. HCV titers were determined using a focus-forming assay as previously described.27,28 Antigen-positive cells were stained with anti-HCV anti-serum and horseradish peroxidase-conjugated goat antihuman IgG (MBL, Tokyo, Japan). Infectious foci were visualized using the Metal Enhanced DAB Substrate Kit (Pierce-Thermo Scientific). Foci in each well were counted and normalized to untreated controls to calculate the titre of virus.
Evaluation of anti-viral activities
Huh7it-1 cells (5.0×104 cells/well) which cultured into a 48-well plate were infected with HCV at a multiplicity of infection of 0.1 in the presence of A. pauciflorum extracts or fractions (100, 50, 20, 10, 5, and 1 μg/ml) for 2 h at 37°C. After removing the residual viruses by rinsing the wells twice with serum-free medium, the cells were incubated for 46 h at the same sample concentrations. At 48 h p.i., the culture supernatants were collected and used for viral titration. In some experiments, the percent infectivity of the fractions at a concentration of 20 μg/ml was determined. Cells were treated with fractions 5 or 13 only during or only after viral inoculation for the remaining culture period until viral harvest to assess the mode of action of the plant extracts.26
Cytotoxicity assay
Cytotoxicities of the crude extracts and the fractions against Huh7it-1 cells were evaluated using the MTT [3-(4,5-dimethylthiazol- 2-yl )-5(3-ca rb ox ymet honyp hen ol )-2-(4- sulfophenyl)-2H-tetrazolium] assay (Sigma- Aldrich) following the manufacturer’s instructions. Cells (2×104 cells/well) were placed in a 96-well plate and treated with a medium containing serial dilutions of the plant extracts or fractions (1 to 300 μg/ml) for 48 h. Cyclosporine A were used as a comparator. The medium was replaced with 100 μl of MTT solution and incubated at 37°C for an additional 4 h. The medium was removed and 100 μl of dimethyl sulfoxide (DMSO, Sigma-Aldrich) was added to dissolve the generated precipitate. Absorbance at 550 nm was measured using a GloMax-Multi Microplate Multimode Reader (Promega). The percentage of viable cells versus the concentration of the test compound was plotted. The concentration by which to mediate 50% inhibition (CC50) was determined by non-linear regression analysis.
Data analysis
The data were presented in mean standard deviation. The data were analyzed using nonlinear regression analysis. IC50 were calculated from the formula of non-linear regression.
RESULTS
Anti-HCV activities of extracts from A. pauciflorum
We examined crude extracts of 250 medicinal plants for anti-HCV effects using a cell-culture HCV system to investigate activities against HCV.24 Huh7it-1 cells, which are highly permissive and obtained by curing replicon cells with IFN-α, were challenged with the HCV genotype 2a strain JFH1 in the presence of extracts. Virus titer in the culture supernatants at 48 h p.i. was determined using a focus-forming assay. Screening results revealed that a crude extract from A. pauciflorum dose-dependently suppressed production of infectious virus particles [50% inhibitory concentration (IC50): 72.5 μg/ml] without cytotoxicity at the concentrations tested. Cytotoxicity of a crude extract to non-infected cells was determined using the MTT assay, which showed a CC50 of 190 μg/ml and a selectively index (SI, CC50/IC50) of 2.6 (Table 1).
Table 1. The effects of methanol crude extract from A. pauciflorum and its fractions on anti-HCV activities (IC50) and cell viabilities (CC50)
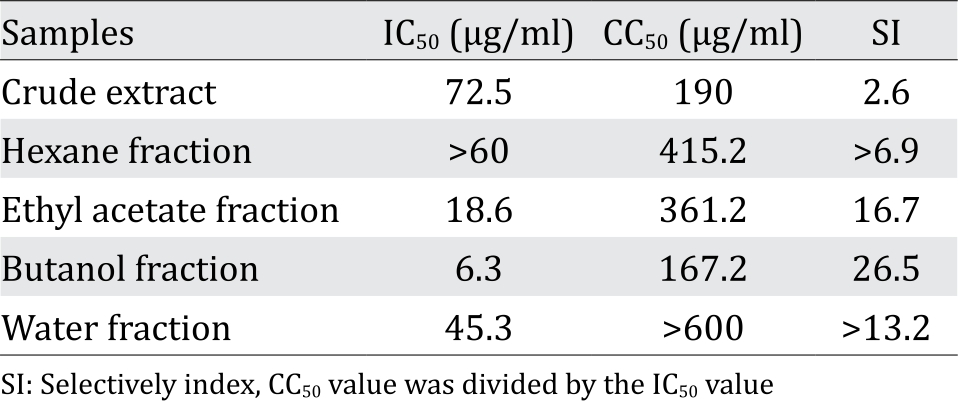
The crude extract was partitioned into n-hexane, ethyl acetate, butanol, and water fractions and subsequently investigated for anti-HCV activities. The butanol fraction had the highest anti-HCV activity with an IC50 value of 6.3 μg/ml (Table 1). The ethyl acetate fraction exhibited moderate anti-HCV activity (IC50, 18.6 μg/ml), whereas the hexane and water fractions exhibited weak or no anti-HCV activities. Cytotoxicities of these fractions to non-infected cells were determined and a CC50 value of the butanol fraction was 167.2 μg/ml, whereas those of other fractions were >300 μg/ml. We selected the butanol fraction for further fractionation due to the high SI value (26.5).
Anti-HCV activities of the isolated fractions
Furthermore, the butanol fraction was separated using silica-based open column chromatograph which yielded 13 fractions as described in figure 2. Cells were infected with HCV in the presence of each fraction (20 μg/ml) and the effects of each fraction on HCV infection were examined. All fraction exhibited varying levels of inhibition of HCV infection. However, we observed that fraction 5 and 13 exhibited a strong inhibition of 100% and 91%, respectively, of the untreated control (Table 2). The IC50 and CC50 values of fraction 5 were 5.0 and 33.2 μg/ml, respectively, whereas those of fraction 13 were 8.5 and 62 μg/ml respectively.
Table 2. Effects of the butanol fractions from A. pauciflorum on HCV JFH1 and cell viabilities
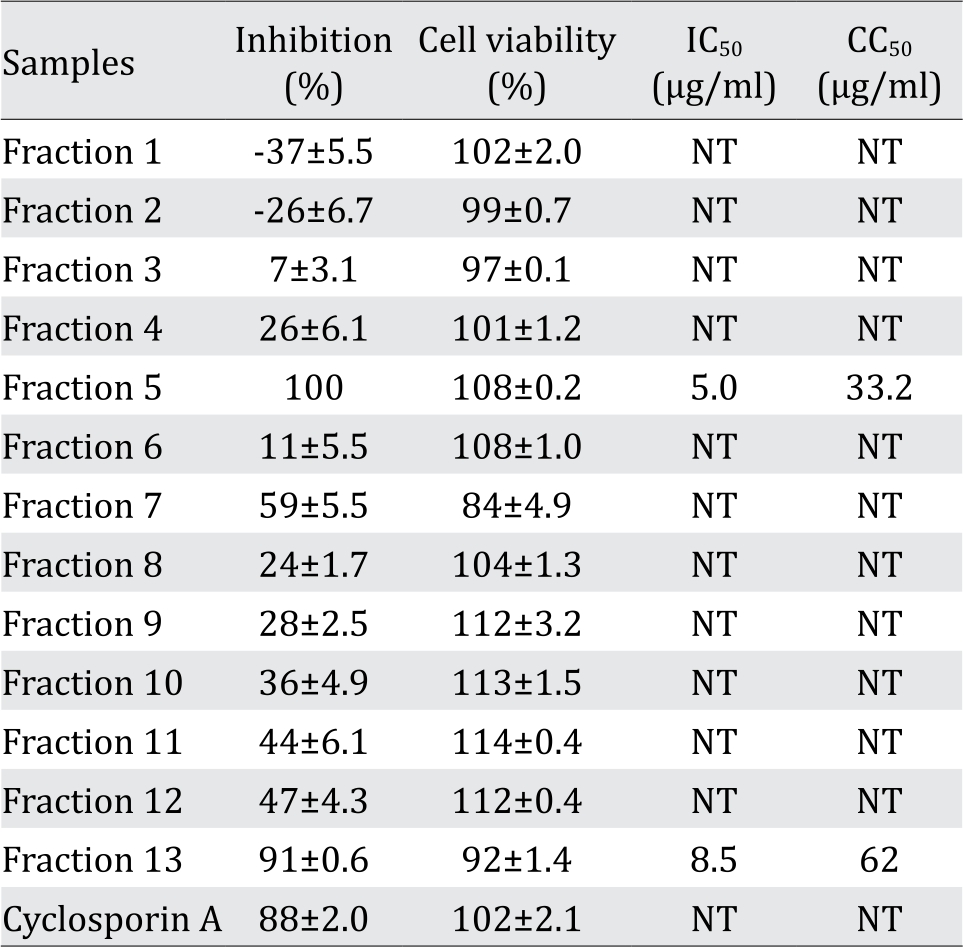
Inhibition and cell viability at the concentration of 20 ug/ml were shown except for the concentration of cyclosporin A as an inhibitor control, which is 1 ug/ml. Values were shown as mean ±SD values of two independent experiments; NT= not tested
Mode-of-action of butanol fraction 5 and 13
To determine whether the anti-HCV effects of the two fraction were inhibited at the entry or the post-entry step, time-of-addition experiments were performed. As presented in Table 3, fraction 5 exhibited anti-HCV activity at the post-entry step, whereas fraction 13 exhibited anti-HCV activity primarily at the entry step. These findings suggested that each fraction contained different compounds with different mechanism of action.
Table 3. Mode of action studies of butanol fractions 5 and 13 from A. pauciflorum
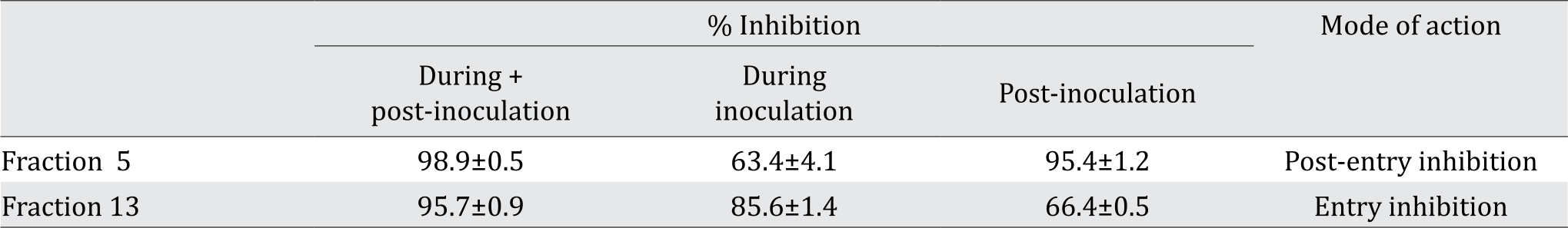
Fractions 5 and 13 at the concentration of 10 μg/ml and 20 μg/ml, respectively. Values presented the mean ± SD from three independent experiments
The percent infectivity by treatment with fraction 5 was plotted for each fixed concentration of fraction 13 (Figure 1a). The curves shifted to the left with increasing concentrations of fraction 13, demonstrating a synergistic effect of the two fraction against HCV infection. This synergistic effect was further evaluated by classic isobologram analysis and the dose-effect plots were drawn. The results demonstrated that the curve was below the line showing an additive effect, indicating that the effect of a combination of the two fractions on HCV infection was strongly synergistic (Figure 1b). Cytotoxicity was not observed at the tested concentrations via isobologram analysis (data not shown). These results collectively suggested that the strong anti-viral activities from the butanol fraction of A. pauciflorum leaves extracts have been contributed to several compounds with different anti-viral mechanisms.
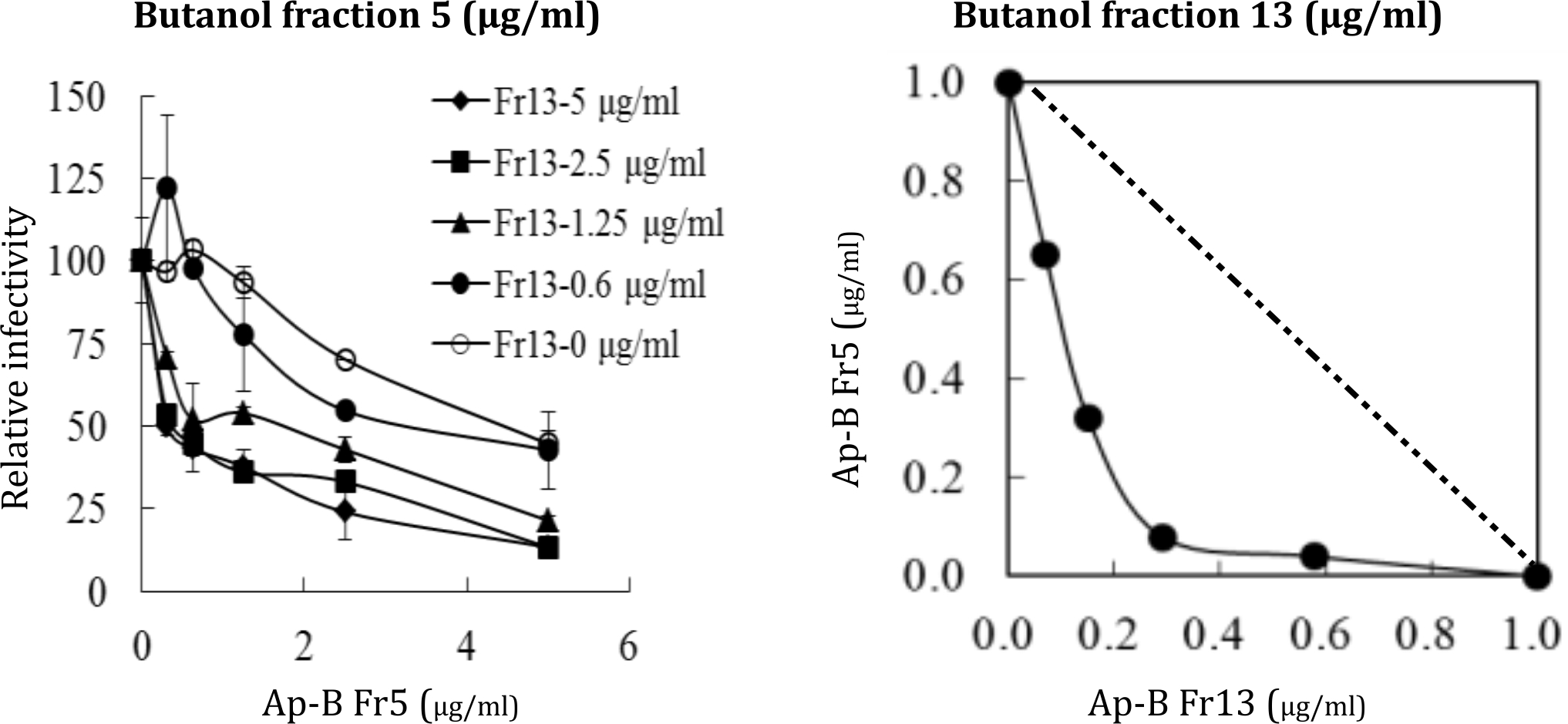
Figure 1. Synergetic effects of the butanol fraction 5 and 13
DISCUSSION
Due to the high rate of pre-existing or emerging viral resistance, effective treatment of HCV-infected patients will likely require a combination of inhibitors targeting distinct viral or host functions. As described above, we demonstrated that both fractions 5 and 13 exerted inhibition activities with different modes of action. On the other hand, the anti-HCV effect by mono-treatment of each butanol fractions were not stronger than that of treatment with the butanol fraction alone. It was possible that fractions have synergistic mode in butanol fractions than each fraction alone. Therefore, we examined the anti-HCV effects of the two fractions in combination. Cells were infected with HCV and treated with combinations of fractions 5 and 13 at various concentrations.
A. pauciflorum (known as A. jiringa, Pithecellobium jiringa in Malaysia) is a typical plant in Southeast Asia including Indonesia. The bean of this plant contains jenkolic acid, sulfur, vitamin C and E, flavonoid, protein, and fatty acids.21–24 The seed is used traditionally for the treatment of hypertension and diabetes, whereas the old leaves burnt to ashes were used against itching parts.25 This plant was also found a high contain of polyphenolic compounds which has a potent antioxidant activity.27 Although the compounds responsible for anti-HCV activities have not yet been isolated in this study, we observed that fraction 5 contained chlorophyll-related compounds, known to show antioxidant activity as major constituents (obtained from our preliminary study). We isolated a chlorophyll-related compound from another Indonesian plant by activity-guided purification and its compound exhibited anti-HCV activity (manuscript in preparation). Also, different groups previously reported that pheophytin and chlorin e6, which are chlorophyll degradation products, exhibited potential anti-HCV activities. The mechanism is through the inhibition of NS3 protease and probably disruption of the envelope protein, respectively.28,29 The activity of fraction 5 may have contributed to the presence of chlorophyllrelated compounds. Further activity-guided purification and analytical studies are required to explore these anti-HCV substances. We have not done the analysis of contents from butanol fraction 13.
In conclusion, crude extract from A. pauciflorum leaves, and the butanol fraction exhibited great potential as anti-HCV agents. The butanol fraction yielded a higher SI value compared to the crude extracts and other fractions. Among 13 butanol fractions, there were two fractions only fractions exhibited strong antiviral activities. Synergetic effects were observed by a combination treatment of these two fractions. The results of the present investigation provide evidence of the potential of medicinal plants for herbal drug as anti-HCV. However, further study for the use of this fraction in vivo is still need to evaluate its efficacy and toxicity.
Conflicts of Interest
The authors affirm no conflict of interest in this study.
Acknowledgment
We thank to Dr. Takaji Wakita for providing pJFH1. This work was supported in part by grants from the Science and Technology Research Partnership for Sustainable Development (SATREPS) from the Japan Science and Technology Agency (JST) and Japan International Cooperation Agency (JICA).
REFERENCES
- Rosen HR. Clinical practice. Chronic hepatitis C infection. N Engl J Med. 2011;364(25):2429–38.
- Ghany MG, Nelson DR, Strader DB, Thomas DL, Seeff LB. An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the study of liver diseases. Hepatology. 2011;54(4):1433–44.
- Balunas MJ, Kinghorn AD. Drug discovery from medicinal plants. Life Sci. 2005;78(5):431–41.
- Polyak SJ, Morishima C, Shuhart MC, Wang CC, Liu Y, Lee DY. Inhibition of T-cell inflammatory cytokines, hepatocyte NF-kB signaling, and HCV infection by standardized silymarin. Gastroenterology. 2007;132(5):1925–36.
- Polyak SJ, Morishima C, Lohmann V, Pal S, Lee DY, Liu Y, et al. Identification of hepatoprotective flavonolignans from silymarin. Proc Natl Acad Sci U S A. 2010;107(13):5995–9.
- Ciesek S, von Hahn T, Colpitts CC, Schang LM, Friesland M, Steinmann J, et al. The green tea polyphenol, epigallocatechin-3-gallate, inhibits hepatitis C virus entry. Hepatology. 2011;54(6):1947–55.
- Nahmias Y, Goldwasser J, Casali M, van Poll D, Wakita T, Chung RT, et al. Apolipoprotein B-dependent hepatitis C virus secretion is inhibited by the grapefruit flavonoid naringenin. Hepatology. 2008;47(5):1437–45.
- Gonzalez O, Fontanes V, Raychaudhuri S, Loo R, Loo J, Arumugaswami V, et al. The heat shock protein inhibitor quercetin attenuates hepatitis C virus production. Hepatology. 2009;50(6):1756–64.
- Lan K-H, Wang Y-W, Lee W-P, Lan K-L, Tseng S-H, Hung L-R, et al. Multiple effects of honokiol on the life cycle of hepatitis C virus. Liver Int. 2012;32(6):989–97.
- Wu S-F, Lin C-K, Chuang Y-S, Chang F-R, Tseng C-K, Wu Y-C, et al. Anti-hepatitis C virus activity of 3-hydroxy caruilignan C from Swietenia macrophylla stems. J Viral Hepat. 2012;19(5):364–70.
- Suzuki M, Sasaki K, Yoshizaki F, Fujisawa M, Oguchi K, Cyong J-C. Anti-hepatitis C virus effect of citrus unshiu peel and its active ingredient nobiletin. Am J Chin Med. 2005;33(1):87–94.
- Manvar D, Mishra M, Kumar S, Pandey VN. Identification and evaluation of anti hepatitis C virus phytochemicals from Eclipta alba. J Ethnopharmacol. 2012;144(3):545–54.
- Polyak SJ, Oberlies NH, Pecheur E-I, Dahari H, Ferenci P, Pawlotsky J-M. Silymarin for hepatitis C virus infection. Antivir Ther. 2013;18(2):141–7.
- Haid S, Novodomská A, Gentzsch J, Grethe C, Geuenich S, Bankwitz D, et al. A plant-derived flavonoid inhibits entry of all HCV genotypes into human hepatocytes. Gastroenterology. 2012;143(1):213–22.
- Chow HH, Cai Y, Hakim IA, Crowell JA, Shahi F, Brooks CA, et al. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin Cancer Res. 2003;9(9):3312–39.
- Panpipat W, Suttirak W, Chaijan M. Free radical scavenging activity and reducing capacity of five southern Thai indigenous vegetable extracts. Walailak J Sci & Tech. 2010;7(1):51–60.
- Bunawan H, Dusik L, Bunawan SN, Mat Amin N. Botany, traditional uses, phytocemistry and pharmacplogy of Archidendron jiringa : a review. 2013, Global J Pharmacol. 2013;7(4):474–8.
- Bakar RA, Ahmad I, Sulaiman SF. Effect of Pithecellbium jiringa as antimicrobial agent. Bangladesh J Pharmacol. 2012;7(2):131–4.
- Muslim NS, Nassar ZD, Aisha AF, Shafaei A, Idris N, Majid AM. Antiangiogenesis and antioxidant activity of ethanol extracts of Pithecelobium jiringa. BMC Complement Altern Med. 2012;12(210):1–10.
- Tee ES, Noor MI, Azudin MN, Idris K. Nutrient composition of Malaysian foods (4th edn). 1997; Malaysian Food Composition database Programme, Institute for Medical Research, Kuala Lumpur: 68–9.
- Thevathasan OI. Hypoglycaemia induced in fasted cats by aqueous extracts of Pithecellobium jiringa. Med J Malaya. 1972;26(3):217–9.
- Razab R, Abdul AA. Antioxidant from tropical herbs. Nat Prod Commun. 2010;5(3):441–5.
- Wang J, Chen X, Wang W, Zhang Y, Yang Z, Jin Y, et al. Glycyrrhizic acid as the antiviral component of Glycyrrhiza uralensis Fisch against coxsackievirus A16 and enterovirus 71 of hand foot and mouth disease. J Ethnopharmacol. 2013;147(1):114–21.
- Lee J-C, Chen W-C, Wu S-F, Tseng C-K, Chiou C-Y, Chang F-R, et al. Anti-hepatitis C virus activity of Acacia confusa extract via suppressing cyclooxygenase-2. Antiviral Res. 2011;89(1):35–42.
- Chrestani F, Sierakowski MR, de Andrade Uchoa DE, Nozawa C, Sassaki GL, Gorin PA, et al. In vitro antiherpetic and antirotaviral activities of a sulfate prepared from Mimosa scabrella galactomannan. Int J Biol Macromol. 2009;45(5):453–7.
- Apriyanto DR, Aoki C, Hartati S, Hanafi M, Kardono LB, Arsianti A, et al. Anti- hepatitis c virus activity of a crude extract from longan (Dimocarpus longan Lour) leaves. Jpn J Infect Dis. 2016;69(3):213–20.
- Yu L, Aoki C, Shimizu Y, Shimizu K, Hou W, Yagyu F, et al. Development of a simple system for screening antihepatitis C virus drugs utilizing mutants capable of vigorous replication. J Virol Methods. 2010;169(2):380–4.
- Aoki C, Hidari KI, Itonori S, Yamada A, Takahashi N, Kasama T, et al. Identification and characterization of carbohydrate molecules in mammalian cells recognized by dengue virus type 2. J Biochem. 2006;139(3):607–14.
- Norulaini NA, Zaidul IS, Azizi CY, Zhari I, Noramin MN, Sahena F, et al. Supercritical carbon dioxide fractionation of Pithecellobium jiringan jack seed compositions using fast gas chromatography time of flight mass spectrometry. J Food Process Eng. 2010;34(5):1746–58.
- Delgado-Vargas F, López-Valdés HE, Valdés-Rodríguez S, Blanco-Labra A, Chagolla-López A, López-Valenzuela EJ. Isolation and properties of kunitz-type protein inhibitor obtained from Pithecellobium dulce seeds. J Agric Food Chem. 2004;52(20):6115–21.
- Wang S-Y, Tseng C-P, Tsai K-C, Lin C-F, Wen C-Y, Tsay H-S, et al. Bioactivity-guided screening identifies pheophytin a as a potent anti-hepatitis C virus.compound from Lonicera hypoglauca Miq. Biochem Biophys Res Commun. 2009;385(2):230–5.
- Guo H, Pan X, Mao R, Zhang X, Wang L, Lu X, et al. Alkylated porphyrins have broad antiviral activity against hepadnaviruses, flaviviruses, filoviruses, and arenaviruses. Antimicrob Agents Chemother. 2011;55(2):478–86.
Copyright @ 2018 Authors. This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are properly cited.
mji.ui.ac.id